What You Should Know:
Flatiron Health today announced its renewed collaboration with the Oncology Center of Excellence (OCE) of FDA to jointly develop and implement specific research projects to advance the use of Real World Data (RWD) and explore the potential strengths and limitations of using Real World Evidence (RWE) for regulatory purposes.The partnership will specifically evaluate RWD study designs and analytic methods through the collaborative development of priority, clinically
Read More
Real World Evidence (RWE)
Surgo Health Launches RWD-Driven Socio-Behavioral Analytics Platform
What You Should Know:
- Surgo Health, a new healthcare technology company dedicated to personalized care announced its official launch in Washington, D.C.
- Formed as a public benefits company (PBC), Surgo Health is building on the analytics and proprietary data assets from Surgo Ventures and is developing a revolutionary socio-behavioral analytics platform and generating novel, real-world data to enable a greater understanding of the complex factors that influence how individuals engage
Read More
Aetion, FDA to Evaluate Real-World Data Standardization for COVID-19
What You Should Know:
- Aetion, a provider of real-world evidence (RWE) technology and analytics, today announced that the FDA has extended Aetion’s contract to evaluate real-world data (RWD) involving COVID-19 medical countermeasures.
- Aetion will be expanding its work to evaluate data standardization in the context of COVID-19. Aetion has been collaborating with FDA since May 2020, providing validated software platform capabilities and RWD/E expertise to contribute to rapid evidence
Read More
Innovaccer Launches Connected Digital Health Solution for Biopharma & Medtech
What You Should Know:
- Today Innovaccer, the Health Cloud company, announced its Connected Digital Health solution, a new way for life sciences companies to rapidly deliver next-generation digital apps that surround therapeutics and devices with highly engaging patient experiences, improve clinical outcomes, and generate valuable real-world evidence (RWE).
- The new solution will allow life sciences companies to realize the promise of digital health with holistic, “around-the-brand”
Read More
Tempus Launches Psychiatric Real-World Data Collaboration
What You Should Know:
- Tempus, an $8B precision medicine company, announced the launch of a new psychiatric real-world data program to leverage real-world data to inform mental health treatment.
- Tempus N+ is a collaboration between provider institutions across the country and Tempus to build an infrastructure for data-driven research – providers can seamlessly and securely contribute molecular and clinical data, and in turn, their clinicians and researchers can access a
Read More
Using NLP Technology To Uncover Real-World Patient Insights
It’s estimated that humans create a combined 2.5 quintillion bytes of data each day – and that around 90% of all data to ever exist was created in just the last few years. As difficult as it is to ignore the massive impact that connected technologies and social media have had on the way we experience our world, it’s somehow even more challenging to fully grasp the scale of it. For health researchers, this data explosion – and especially the rise
Read More
ConcertAI Raises $150M at $1.9 Billion Valuation for RWD Platform
What You Should Know:
- ConcertAI, LLC (ConcertAI), the leader in enterprise AI and real-world data (RWD) solutions for life sciences and healthcare, announced the close of its $150M in Series C led by Sixth Street at a $1.9B valuation.
- From its startup just five years ago, ConcertAI has emerged as one of the fastest-growing companies providing high-value software-as-a-service (SaaS) and data solutions to life sciences and biopharma companies, leading healthcare providers, contract
Read More
Implementing Responsible AI to Drive Innovation in Clinical Research & Precision Medicine
Researchers might be skeptical and, with people’s lives at stake, that’s understandable. Here’s how to take advantage of the advances while ensuring safeguards are in place.
As artificial intelligence (AI) and machine learning drive innovation and disruption in clinical trials and precision medicine, questions around the responsible use of AI have moved to the forefront of debates among healthcare stakeholders – patients, providers, regulatory authorities, payors and sponsors. To what extent,
Read More
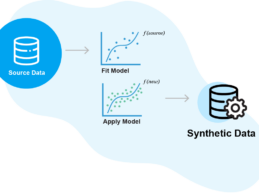
Aetion Acquires Synthetic Data Platform Replica Analytics
What You Should Know:
- Aetion, a real-world evidence (RWE) tech platform, has just announced its acquisition of Replica Analytics, a pioneer in using ML to generate synthetic data. Financial details of the acquisition were not disclosed.
- This acquisition allows Aetion to solve for one of the toughest problems in RWE: access to fit-for-purpose data. Synthetic data acts as a “digital clone” of real-world data by maintaining the source data’s statistical properties, while also protecting
Read More
Using Real-World Evidence (RWE) & AI to Enable Better Clinical Care
During the prescribing process, a physician’s ability to accurately and reliably match the most appropriate therapy option with the patient at hand is limited by the amount and quality of clinical information that is available. While randomized clinical trials are considered the gold standard for establishing the safety and efficacy of prescription medications, and the clinical findings verified from the trial inform the label on the approved therapy, the overall utility of such trials is
Read More