HealthQuest Capital, a Silicon Valley-based growth capital firm has closed a $440 million fund to invest growth-stage companies delivering innovation across all sectors of the healthcare industry to improve patient outcomes, enhance efficiency and reduce costs. Fund III was oversubscribed and closed at a hard cap. Target markets for Fund III include growth- and commercial-stage companies focusing on medical technologies, diagnostics, digital health, and innovative services.HealthQuest Capital
Read More
Medical Devices | Policy & Regulation News, Analysis, Insights - HIT Consultant
3M Unveils Medical Transfer Adhesive for Wearable Medical Devices
Today, 3M announced it has added 3M Extended Wear Medical Transfer Adhesive, 4075 to its lineup of advanced adhesives for wearable medical devices. Designed to laminate many substrates, 3M medical transfer adhesive, 4075 features an extended wear pressure sensitive transfer adhesive and allows design engineers to use a variety of backings. 3M medical transfer adhesive, 4075 offers excellent initial skin adhesion with up to a 14-day wear time, depending on the backing material used.The Critical
Read More
6 Patient Influencers Redefining The Healthcare Landscape
Most people are now familiar with social media influencers. They're the individuals who use their popularity to encourage followers to try clothing brands, food, makeup, and gadgets. As anyone who watched either of the two Fyre Festival documentaries now knows, they can be instrumental in getting people on board with things associated with disastrous consequences.
There's a relatively new concept in the influencer world. It involves people known as patient influencers doing just as the name
Read More
HIMSS19 Day 2 Announcements & Summary
Accenture: Millenials & Gen Z Are Shifting to Virtual, Retail, Clinic, Digital CareMillennial and Gen Z consumers in the U.S. are rapidly adopting non-traditional care models, such as retail clinics, virtual and digital services, according to results of an Accenture survey of 2,000 consumers.Key findings of the survey study include:- When considering traditional in-person care, millennials (ages 22 to 38 in 2019) were two to three times more likely than baby boomers (ages 55 to 73) to be
Read More
Francisco Partners Acquires Qualcomm Life, Renames as CapsuleTech
Francisco Partners ("FP"), a Silicon Valley-based technology-focused private equity (PE) firm, today announced it has acquired Qualcomm Life, Inc. a wholly owned subsidiary of Qualcomm Incorporated that offers end-to-end medical device connectivity across the continuum of care. Financial details of the acquisition were not disclosed. Renames Qualcomm Life as CapsuleTechQualcomm Life will be separated from Qualcomm, be renamed as Capsule Technologies, Inc. ("CapsuleTech"), and continue operating
Read More
Why the FDA Is Doing an Overhaul of the Medical Device Approval Process
Since 1976, the U.S. Food and Drug Administration (FDA) has allowed manufacturers to apply for an accelerated pathway to bringing their devices to the market. It's called the 501(k) Premarket Notification. In short, if a new device gets proved safe and effective to a device that's already available, the new device can get cleared because it's considered substantially similar to the older one. The FDA also allows for the first-cleared device (also known as the predicate) to be another product
Read More
How Will Apple’s New iPad Pro Impact Healthcare at the Point of Care?
DrChrono's Daniel Kivatinos explains how healthcare could benefit from going pro with Apple’s newest iPad.Take a good look at Apple’s iPad Pro, and you may see why the device could open new doors in productivity. In fact-- a glance is all that’s required to unlock it thanks to Face ID, one of its most appealing features. What could be improved if you put Apple’s Pro into the hands of willing healthcare providers?It turns out much good could come from employing the device in healthcare; it has
Read More
Biotech Startup ParaGen Technologies Raises $3M to Redefine Tissue Engineering
ParaGen Technologies, a Columbus, OH-based holding company of four tissue engineering medical device companies, announced that it raised $3 million in funding. The round was managed and organized through Ikove Venture Partners’ global network of investors (70% of the funds came from investors across 10 countries).Tissue EngineeringThe field of tissue engineering is focused on developing solutions to recreate the biological structures of the body. Generally, the synthetic products currently
Read More
BlackBerry Named HIMSS Analytics Certified Consultant to Guides Organizations Through Stages of INFRAM
Today, BlackBerry announced it has become a HIMSS Analytics Certified Consultant to guide healthcare organizations through the stages of the Infrastructure Adoption Model (INFRAM), as well as ERAM. Professionally trained on the INFRAM, BlackBerry’s cybersecurity experts can assess a healthcare organization’s infrastructure architecture and help them achieve their desired INFRAM score.Understanding Infrastructure Adoption Model (INFRAM)Similar to the HIMSS Analytics Electronic Medical Record
Read More
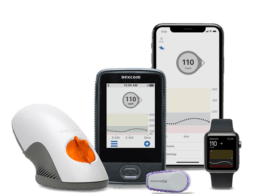
DexCom Integrates Continuous Glucose Monitoring Data with Validic Data Connectivity Platform
DexCom, Inc., a provider of continuous glucose monitoring (CGM) solutions, announced a partnership with data integration platform Validic. As part of the partnership, DexCom will integrate CGM data from Dexcom’s cloud API is integrated into the Validic’s connectivity platform to establish a multi-measure platform to stream in-home clinical data directly into clinical workflows and systems. This partnership is part of a larger effort to improve the interoperability of clinical systems and provide
Read More