What You Should Know:
- Nebraska-based Bluestem Health today announced it has launched an AI-based virtual medical assistant powered by Mediktor. This is available for both established and non-established patients to efficiently assess their symptoms online and direct them to the appropriate level of care.
- Mediktor combines AI, Natural Language Processing (NLP) and machine learning technologies for the first clinically validated digital medical assistant, proven in clinical trials with
Read More
Clinical Trials | News, Analysis, Insights - HIT Consultant
5 Components of a Patient-Centric eCOA Strategy for Oncology Clinical Trials
In the US alone, an estimated 1.9 million new cases of cancer were diagnosed in 2022, positioning oncology as a key subject of clinical research. Throughout oncology trial development, it is important that stakeholders acknowledge that only patients can fully understand the impact of treatment on their lives. Regulators are now looking beyond clinical indications such as tumor size and delayed disease progression. When evaluating the risks and benefits of treatment, they want to know whether the
Read More
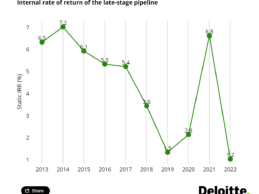
Report: Pharma R&D ROI Falls to Lowest Level in 13 Years
What You Should Know:
- Deloitte Centre for Health Solutions releases its thirteenth annual report, ‘Seize the Digital Momentum: Measuring the return from pharmaceutical innovation 2022’ that explores the performance of the biopharmaceutical industry (bio Pharma) and its ability to generate returns from investment in innovative new medicines.
- Since 2021, much of the world has successfully adjusted to life where COVID-19 is more endemic as vaccines and treatments reduced the risk and
Read More
Faro Health Secures $20M to Streamline Smart Clinical Trials
What You Should Know:
- Faro Health, a cloud-computing company that is developing digital infrastructure for smart clinical trials raises $20M in Series A funding led by General Catalyst to help scale the digital transformation of clinical trials––essentially making it easier and quicker for drug developers to greatly reduce the cost and time of trials.
- The round also included participation from previous investors Section 32, Polaris Partners, Zetta Ventures, and Northpond Ventures.
Read More
Biden Admin Builds on Cancer Moonshot with 13 More Initiatives
What You Should Know:
- One year ago, President Joe Biden and First Lady Jill Biden reignited the Cancer Moonshot, setting an ambitious, achievable goal: to reduce the death rate from cancer by at least 50 percent over the next 25 years, and improve the experience of people and families living with and surviving cancer, ultimately ending cancer as we know it today.
- The Cancer Moonshot has spurred tremendous action across the federal government and from the public and private
Read More
2023’s Biggest Pharma Cybersecurity Threats to Watch
Due to the recent growth of the pharmaceutical industry, the value of highly-sensitive data stored in pharmaceutical systems and the degree of the potential damage that cyberattacks on the industry can cause, it is safe to say that pharma could be one of the most targeted industries by cybercriminals in 2023. Ransomware, phishing attacks, business applications and third-party vendors will be some of the biggest threats to this key vertical as we approach the new year.
RansomwareThe threat of
Read More
ProKidney Taps CVS Health Clinical Trial Services for Precision Recruitment
What You Should Know:
- CVS Health Clinical Trial Services and late clinical-stage cellular therapeutics company ProKidney Corp. today announced a collaboration to enroll patients with type 2 diabetes and chronic kidney disease into the proact 1 (REGEN-006) Phase 3 clinical trial of REACT®.
ProKidney’s lead product candidate, REACT® (REnal Autologous Cell Therapy), is a first-of-its-kind, patented, autologous cellular therapy with the potential to not only slow and stabilize the
Read More
Curebase Launches Remote First DTx Research Platform
What You Should Know:
- Curebase, a company committed to democratizing access to clinical trials, announced the release of its integrated software and services package for digital therapeutics (DTx) trials that accelerates enrollment and allows sponsors to launch their studies faster.
- Available now, the new Curebase offering includes the first one-stop-shop decentralized clinical trial (DCT) platform bundled with site and trial execution services for the DTx market.
Making
Read More
Healthcare AI Transformation: 9 Executive Predictions to Watch in 2023
Piotr Orzechowski, Co-Founder and CEO of Infermedica
Innovation areas to watch: One of the exciting areas of applied machine learning recently is so-called Generative Tech. Some say it’s the biggest change to the Internet since crypto was invented. Generative Tech is developing at an unprecedented pace and many of us already had a chance to enjoy images created by OpenAI’s DALLE-E or content generated by GPT-3 language model. However real-world applications of generative tech in health are
Read More
Boosting Clinical Trial Recruitment Requires Humanity – and Technology
Clinical trials should be a time of promise for better patient outcomes, as they explore new ways to potentially help patients suffering from a variety of conditions. Instead, this phase of treatment development is often met with exasperation as patients, researchers, and drug developers deal with the many barriers to clinical trial recruitment and access.
There are two central – and intrinsically linked – problems with the current state of clinical trial recruitment in the U.S.:
Read More