What You Should Know:
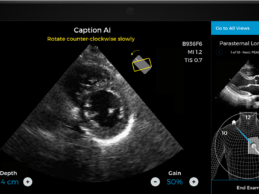
- Caption Health AI is awarded FDA 510(k) clearance for
its innovative point-of-care ejection fraction evaluation.
- Latest AI ultrasound tool makes it even easier to
automatically assess ejection fraction, a key indicator of cardiac function, at
the bedside--including on the front lines of the COVID-19 pandemic.
Caption Health, a Brisbane,
CA-based leader in medical AI technology, today announced it has received FDA
510(k) clearance for an updated version of
Read More
FDA clearance 510k
Zebra Medical Vision Secures 5th FDA Clearance for Vertebral Compression Fractures AI Solution
What You Should Know:
- Israeli deep-learning medical imaging analytics startup Zebra-Med secures its fifth FDA clearance vertebral compression fractures AI solution for availability in the U.S.
- The AI solution will help health providers close the COVID-19-induced care gaps by identifying more patients at risk of osteoporosis, also known as the “silent killer,” costing over $52B in the U.S. alone.
- Zebra-Med is one of the first companies in the industry to provide an AI
Read More
FDA Clears AI-Powered EchoGo Core for Early Detection of Cardiovascular Disease
- Ultromics has just become one of the first companies to have 510(k) FDA clearance for an AI medical device. - EchoGo will now be available for clinicians in the UK and US to use to help them identify cardiovascular disease earlier. - EchoGo automates cardiac measurements and is the first AI application to measure cardiac strain, improving patient care and outcomes.Ultromics, the UK-based health technology firm at the forefront of applying artificial intelligence to echocardiography has
Read More
Biofourmis Analytics Engine Receives FDA Clearance for Ambulatory Physiologic Monitoring
- Biofourmis received 510(k) clearance from the FDA for its Analytics Engine as a medical device for ambulatory physiological monitoring.- Milestone approval establishes an AI-powered solution as the foundation for future disease-specific predictive management tools from Biofourmis.- This FDA approval is the second market authorization for Biofourmis, having earned the agency's approval in May 2019 for its Biovitals RhythmAnalytics platform.Boston-based Biofourmis has received 510(K) clearance
Read More
FDA Clears GE Healthcare’s AI Algorithms Embedded on Mobile X-Ray Device
- GE Healthcare awarded Food and Drug Administration’s 510(k) clearance of Critical Care Suite, an industry-first collection of artificial intelligence (AI) algorithms embedded on a mobile X-ray device.
- Algorithms help radiologists prioritize critical cases with a suspected pneumothorax – a type of collapsed lung – by immediately flagging critical cases to radiologists for triage, which could drastically cut the average review time from up to eight hours.
- Critical Care Suite offers
Read More
Medivis Granted FDA 510(k) Clearance for Surgical Augmented Reality Platform
Medivis, a New York City-based medical technology company harnessing augmented reality and artificial intelligence to advance surgical visualization has received 510(k) clearance for clinical use in the operating room by the U.S. Food and Drug Administration (FDA).Most medical procedures are performed relatively blindly, with surgeons having to reconstruct slices of 2D imaging data in the “mind’s eye” to make it actionable. Founded by neurosurgeon Osamah Choudhry, MD and radiologist Christopher
Read More
Why the FDA Is Doing an Overhaul of the Medical Device Approval Process
Since 1976, the U.S. Food and Drug Administration (FDA) has allowed manufacturers to apply for an accelerated pathway to bringing their devices to the market. It's called the 501(k) Premarket Notification. In short, if a new device gets proved safe and effective to a device that's already available, the new device can get cleared because it's considered substantially similar to the older one. The FDA also allows for the first-cleared device (also known as the predicate) to be another product
Read More
New Philips App Enhances Mobility for Caregivers with Real-Time Access to Patient Deterioration
Philips today announced it has expanded its General Care Solution with the new Philips IntelliVue GuardianSoftware mobile client application. The mobile app has received 510(k) clearance from the FDA and leverages the capabilities of Philips’ General Care solution, which includes Early Warning Scoring (EWS), to provide clinicians with the benefit of mobility and optimized workflows. In some hospital medical-surgical units, patient information and color-coded early warning scores are written on a
Read More
University Hospitals Adopts Needle-Free Blood Draw Technology
University Hospitals Cleveland Medical Center adopts needle-free blood draw technology to reduce anxiety and risks for hospitalized patients.University Hospitals (UH) Cleveland Medical Center in Ohio, announced that it has implemented the PIVO needle-free blood draw device from San Francisco-based medical device innovator Velano Vascular at UH Cleveland Medical Center for inpatient blood draws. UH is the first health system in Ohio to deliver this enhanced experience to patients and is helping
Read More
DarioHealth Awarded FDA (510K) Clearance for iPhone Smart Glucose Meter App
DarioHealth, an Israli-based global digital health company with mobile healthand big data solutions, announced today that the United States Food and Drug Administration (FDA) has granted Pre-market Notification (510(k)) clearance for the Company's Lightning®-enabled version of the acclaimed Dario Blood Glucose Monitoring System which enables the use of the Dario app on iPhone 7, 8 and X smart mobile devices (SMD). Consumers in the U.S. market will be able to receive the same quality user
Read More