What You Should Know:
- Philips receives 510(k) clearance from the FDA for its pre-hospital
wireless monitoring solution (Tempus LS- Manual), now offering its remote
monitor and defibrillator solution (Tempus ALS) to EMS customers in the U.S.
- Solution delivers real-time bidirectional data transfer
for remote patient monitoring, giving EMS responders a new approach to
pre-hospital care.
Philips, today announced the launch of its
remote monitoring and defibrillator solution
Read More
FDA| Healthcare FDA Regulation | Policy, News, Analysis, Insights - HIT Consultant
Ciox Health Acquires Biomedical NLP Company Medal
What You Should Know:
- Ciox Health has acquired Medal, Inc., a biomedical
Natural Language Processing (biomed-NLP) technology company.
- The acquisition will lead Ciox to better and more
quickly enable real-world data in support of research that advances patient
care.
Ciox
Health, a leading health technology company, today announced its acquisition
of San Francisco-based biomedical Natural Language Processing (biomed-NLP)
technology company, Medal, Inc., a leader
in
Read More
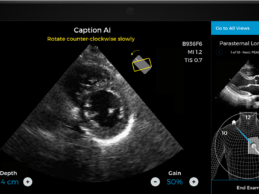
Caption Health AI Awarded FDA Clearance for Point-of-Care Ejection Fraction Evaluation
What You Should Know:
- Caption Health AI is awarded FDA 510(k) clearance for
its innovative point-of-care ejection fraction evaluation.
- Latest AI ultrasound tool makes it even easier to
automatically assess ejection fraction, a key indicator of cardiac function, at
the bedside--including on the front lines of the COVID-19 pandemic.
Caption Health, a Brisbane,
CA-based leader in medical AI technology, today announced it has received FDA
510(k) clearance for an updated version of
Read More
Call to Action: Restrictions Around Virtual Health Can’t Go Back to “Normal”
As a result of the COVID-19 pandemic, the federal government has passed several stimulus packages with the goal of helping employers keep their business in operation while office volume is down and, in some cases, practices are physically closed. The CARES Act, which included the Paycheck Protection Program (PPP) and Provider Relief Fund, for example, has provided some relief to eligible healthcare providers, including low-interest rate loans to help providers pay staff salaries, rent, and other
Read More
Eko Awarded $2.7M NIH Grant for Heart Murmur & Valvular Heart Disease Detection Algorithms
What You Should Know:
- The National Institutes of Health (NIH) has granted next-generation
cardiac AI company Eko an award totaling $2.7 million to support continued
collaborative work with Northwestern Medicine Bluhm Cardiovascular Institute
- The grant will focus on validating algorithms and help
more accurately screen for heart murmurs and valvular heart disease during
routine office visits with Northwestern Medicine.
- By incorporating data from tens of thousands of
Read More
Activ Surgical Raises $15M Advance Autonomous and Collaborative Surgery
What You Should Know:
- Digital surgery pioneer Activ Surgical raises $15M in funding led by ARTIS Ventures to accelerate U.S. commercialization and European expansion efforts for its ActiveEdge platform, which enables existing surgical systems, from scopes to robots, to visualize, characterize and track tissue, in real-time beyond today’s human capability.
- ActivSight, is an easy-to-adapt, connected imaging module that seamlessly attaches to today’s laparoscopic and arthroscopic
Read More
Caption Health Nabs $53M to Commercialize FDA-Cleared AI-Guided Ultrasound Technology
What You Should Know:
- Caption Health raises $53 million series B round to
fuel commercialization and expansion of its FDA-approved AI ultrasound
technology. This is on the heels of its receiving FDA clearance, designated as
a breakthrough technology to fight against COVID-19.
- As ultrasounds, especially cardiac ultrasounds, have
become increasingly important during a time of COVID when resources are
stretched, using AI to help more healthcare professionals take high-quality
images is
Read More
Scripps Research, Repurpose.AI Partner to Develop COVID-19 Therapeutics
What You Should Know:
- Repurpose.AI, has partnered with Scripps Research to
discover drug candidates that may be repurposed to treat COVID-19.
- The partnership will leverage Repurpose.AI's ActivPred
AI Drug Discovery Platform, an unbiased drug, target, and disease agnostic
digital chemistry engine, to discover drug candidates to treat COVID-19.
Scripps Research is
teaming up with Repurpose.AI, an AI
drug discovery company to develop COVID-19 therapeutics. The
Read More
Philips and BioIntelliSense Integrate to Enhance Remote Patient Monitoring
What You Should Know:
- Philips integrates the BioIntelliSense FDA-cleared
BioSticker™ sensor as part of its remote patient monitoring solutions for
patients outside the hospital.
- Multi-parameter sensors aid monitoring across multiple chronic conditions with medical-grade vital signs for physicians to remotely track core symptoms, including COVID-19.
- Healthcare Highways is the first to leverage the BioSticker sensor as a part of Philips’ RPM program in the U.S.
Philips,
Read More
18 Healthcare Augmented Reality and Virtual Reality Companies to Watch
Augmented reality (AR) and virtual reality (VR) are redefining what's possible in healthcare, as well as other sectors. The 18 companies listed below are some of the top virtual and augmented reality companies in the medical industry.
Defining Augmented Reality
Augmented
reality is a technology that blends real-world elements with virtual ones. For
example, a person might visit a trade show and aim their smartphone at a
display to activate an AR experience that allows them to see
Read More