Syqe Medical, an Israeli pharma-tech company, recently launched its newest administering device for cannabis in the form of a non-combustion inhaler.
The Syqe Inhaler™ is the company’s flagship product that utilizes innovative technology to administer proper doses of cannabis for medical marijuana patients in Israel. Syqe’s breakthrough drug delivery technology introduces complex respiration technique automation, electronic selective dosing, remote clinical monitoring and dose
Read More
Medical Devices | FDA | News, Analysis, Insights - HIT Consultant
NextGate Integrates EMPI with Life Image’s Clinical & Imaging Data Network
NextGate, a provider of healthcare enterprise identification, and Life Image, a global network for sharing clinical and imaging data that is powered by industry leading interoperability standards, today announced a strategic partnership to meet the increasing demand for reliable imaging workflow integration.Global-Sharing Network of Clinical, Imaging, Medical Device and Pharmaceutical DataEliminating silos and ensuring that each patient has one and only one record, NextGate and Life Image will
Read More
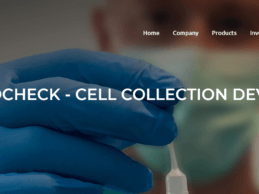
Lucid Diagnostics Receives FDA 510(k) Clearance for EsoCheck
PAVmed Inc. (the “Company” or “PAVmed”), a highly differentiated, multi-product medical device company, today announced that its majority owned subsidiary, Lucid Diagnostics Inc. (“Lucid”), has received 510(k) marketing clearance for its EsoCheck Cell Collection Device™ (“EsoCheck”) from the U.S. Food and Drug Administration (FDA).
EsoCheck is a non-invasive cell collection device that is designed to sample cells from a targeted region of the esophagus in a five-minute office-based
Read More
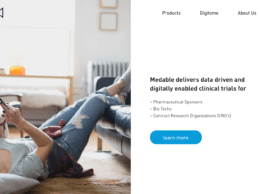
Medable Launches Telemedicine Solution to Improve Data-Driven Virtual Clinical Trials
Medable Inc., a Silicon Valley-based provider of data driven and digitally enabled clinical trials has announced the launch of their global Trial-Fit™ Telemedicine Solution designed to address critical trial barriers of low patient retention and lack of access. Medable’s Trial-Fit Telemedicine enables sites all over the world to conduct secure, HIPAA-compliant video consultations with clinical study participants in an integrated eCOA solution. Medable telemedicine is the only integrated,
Read More
Dassault Systèmes Acquires Clinical Trial Platform Medidata for $5.8B
French technology giant Dassault Systèmes has agreed to acquire Medidata, a life sciences digital transformation company in an all-cash deal for $92.25 per share valued at $5.8 billion. With the acquisition of Medidata and its clinical and commercial solutions, Dassault Systèmes plans to increase market penetration into the Life Sciences industry with an integrated business experience platform for an end-to-end approach to research and discovery, development, clinical testing, manufacturing and
Read More
2019 MedTech Breakthrough Award Category Winners Announced
MedTech Breakthrough, a market intelligence organization that recognizes the top companies, technologies, and products in the global digital health and medical technology market has unveiled the results of its 2019 MedTech Breakthrough Awards program, showcasing technologies and companies that drive innovation and exemplify the best in medical and health-related technology solutions across the globe.MedTech Breakthrough Awards Program OverviewThe MedTech Breakthrough Awards program is the
Read More
Flow Launches Medical-Grade Headset, Therapy App for Depression
Flow, a medical device company launches a medication-free treatment for depression comprising a brain stimulation headset and therapy app. In Europe, Flow has been approved as a Class II medical device intended for use as a treatment for depression - and is the first approved treatment of its kind in Europe available to buy and use at home.Flow BackgroundBased in Sweden, Flow was developed by clinical psychologist Daniel Mansson and neuroscientist Erik Rehn, together with a team of prominent
Read More
Pillo Health Raises $11M to Expand Personal Home Health Robot
Pillo Health, a Boston-based digital health company improving longevity and quality of life for patients managing their health at home has raised $11M in Series A funding, led by Stanley Black & Decker’s corporate venture capital arm with significant participation from Samsung Ventures. Existing investors, including BioAdvance, Hikma Ventures – the corporate venture capital arm of Hikma Pharmaceuticals PLC – Hackensack Meridien Health System’s Innovation Center Fund and
Read More
RSIP Vision Launches AI-Driven Solution for Knee Replacement Patients
RSIP Vision, a provider artificial intelligence (AI), computer vision, and image processing technology, has announced the release of a new AI module that promises a life-changing impact on the millions of patients who undergo knee surgery every year. For most, knee replacement surgery is a difficult option, with a painful recovery and a long, rigorous rehabilitation period. Now the superior accuracy and precision of the RSIP Vision AI solution, unmatched in the market today, revolutionizes knee
Read More
3 Major Problems With the Medical Device and Wearables Market in 2019
Technology can improve human life in significant ways and aid in the fight against troublesome illnesses. Unfortunately, no technology is perfect — and newer technologies are especially prone to growing pains.There are three major problems in today's medical devices and wearables market: potential sabotage of the devices themselves, devices as a backdoor into networks and patient data, and device manufacturers taking advantage of regulatory loopholes to get their products on the market.1. Many
Read More