What You Should Know:
- Scopio Labs, a leader in digital cell morphology, has achieved a major breakthrough with FDA De Novo clearance for its Full-Field Bone Marrow Aspirate (FF-BMA) Application.
- The FDA clearance marks a pivotal moment, establishing the first-ever regulatory category for digital bone marrow aspirate analysis software in the US.
Bone Marrow Analysis: Crucial for Diagnosing Blood Disorders
Microscopic analysis of bone marrow samples, known as bone
Read More
FDA Clearance
Dexcom Earns FDA Clearance for OTC Glucose Biosensor in the U.S.
What You Should Know:
DexCom, Inc. (NASDAQ:DXCM), the global leader in real-time continuous glucose monitoring for people with diabetes, announced today that the FDA has cleared Stelo by Dexcom – the first glucose biosensor that doesn’t require a prescription.There are approximately 25 million people in the U.S. living with Type 2 diabetes who do not use insulin and who can benefit from continuous glucose monitoring (CGM) technology. Today, Dexcom G7 is available for them with a
Read More
X-trodes’ Smart Skin Earns FDA Clearance for Home-Based Electrophysiological Monitoring
What You Should Know:
- X-trodes, a pioneer in wireless monitoring solutions, has achieved a major milestone with the U.S. Food and Drug Administration (FDA) granting 510(k) clearance for its Smart Skin solution, marketed as the X-trodes System M.
- The FDA clearance positions X-trodes as a frontrunner in democratizing advanced electrophysiological monitoring. By bringing clinical-grade technology to the home environment, X-trodes paves the way for personalized healthcare solutions,
Read More
23andMe Scores FDA Clearance for Novel Cancer Immunotherapy Targeting NK Cells
What You Should Know:
- 23andMe Holding Co. (Nasdaq: ME), a genetics and biopharmaceutical company, announced The U.S. Food and Drug Administration (FDA) has cleared the investigational new drug (IND) application for 23ME-01473 (referred to as '1473), paving the way for the first human clinical trial of this promising new therapy.
- With FDA clearance secured, 23andMe plans to initiate a Phase 1 clinical trial in the first half of 2024. This initial study will evaluate the safety and
Read More
AI Breakthrough for Lung Disease: Thirona’s LungQ Receives FDA Clearance
What You Should Know:
- Thirona, a pioneer in AI-powered lung analysis, received a major boost for its cutting-edge technology LungQ™ with the U.S. Food and Drug Administration (FDA) 510(k) clearance for its latest update, v3.0.0.
- This FDA clearance paves the way for American hospitals to now harness the software's advanced capabilities, marking a significant step forward in lung disease diagnosis and treatment.
LungQ 3.0.0: Precision Navigation for Lung Interventions
This latest
Read More
Smileyscope Receives FDA Clearance for First-Ever VR Analgesic
What You Should Know:
- Smileyscope™, a pioneer in virtual reality (VR) therapeutic solutions, has received Class II clearance from the U.S. Food and Drug Administration (FDA) for its Smileyscope™ Therapy system.
- The FDA clearance marks Smileyscope as the first and only VR Analgesic™ available in the US.
Digital Therapeutics Leadership
Smileyscope's FDA clearance underscores its leadership in Digital Therapeutics, a category defined as delivering medical interventions directly to
Read More
FemTech: Mosie Baby Awarded FDA Clearance for At-Home Intravaginal Insemination
What You Should Know:
Mosie Baby, a pioneering at-home fertility care company, has secured FDA Class II clearance for its Mosie Baby Kit making it the first and only FDA-cleared over-the-counter kit for use in intravaginal insemination (IVI).The kit was created to support those who are unable to conceive with intercourse or for whom intercourse is not an option. Following its FDA 510k Class II clearance, Mosie Baby looks forward to expanding access to its Mosie Baby Kit which was
Read More
RapidAI Receives FDA Clearance for Rapid SDH (Subdural Hematoma) for Trauma Care
What You Should Know:
- RapidAI has received FDA clearance for Rapid SDH, its AI-powered module for the detection and notification of suspected hemispheric acute and chronic subdural hematoma.
- The need for the RapidAI solution is urgent, with SDH cases in US patients expected to increase by nearly 80% before 2040.
AI Module for Detection of Hemispheric Subdural Hematomas
Rapid SDH leverages AI to help neurocritical care teams identify suspected hemispheric subdural hemorrhage
Read More
Empatica Awarded New FDA Clearance for Cardiac Digital Biomarkers
What You Should Know:
- Empatica, a digital health and AI company, has obtained FDA 510(k) clearance for two new digital biomarkers, pulse rate, and respiratory rate, on its Empatica Health Monitoring Platform.
- This clearance expands the platform to include a total of six FDA-cleared digital biomarkers, out of the 128 digital measures supported, making it one of the most comprehensive solutions available for use in clinical trials.
- The Empatica Health Monitoring Platform comprises a
Read More
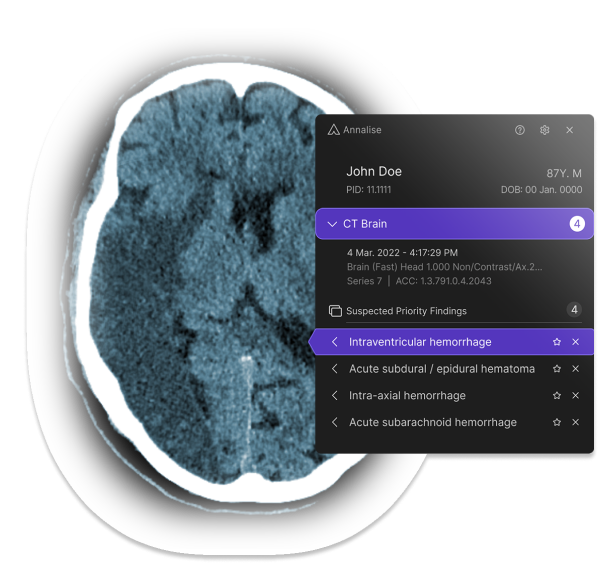
Annalise.ai Receives FDA Clearance for Radiology Triage Device
What You Should Know:
Annalise.ai, a leader in AI-powered medical imaging solutions, announced the receipt of 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the triage and notification of obstructive hydrocephalus (OHCP) on non-contrast brain CT scans.Annalise.ai has also received FDA Breakthrough Device Designation for its obstructive hydrocephalus software tool. This is the first radiology triage device to be granted Breakthrough status since the inception of the
Read More