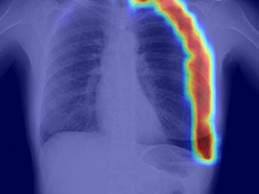
What You Should Know:
- Implicity, a leader in remote patient monitoring and cardiac data management, has received FDA 510(k) clearance for SignalHF1, a groundbreaking new algorithm within their remote monitoring solution.
- SignalHF1 algorithm empowers early detection and intervention, paving the way for improved patient outcomes and a brighter future for cardiac care.
Leveraging Big Data for Better Heart Care
Implicity holds a unique distinction as the first private
Read More
FDA clearance 510k
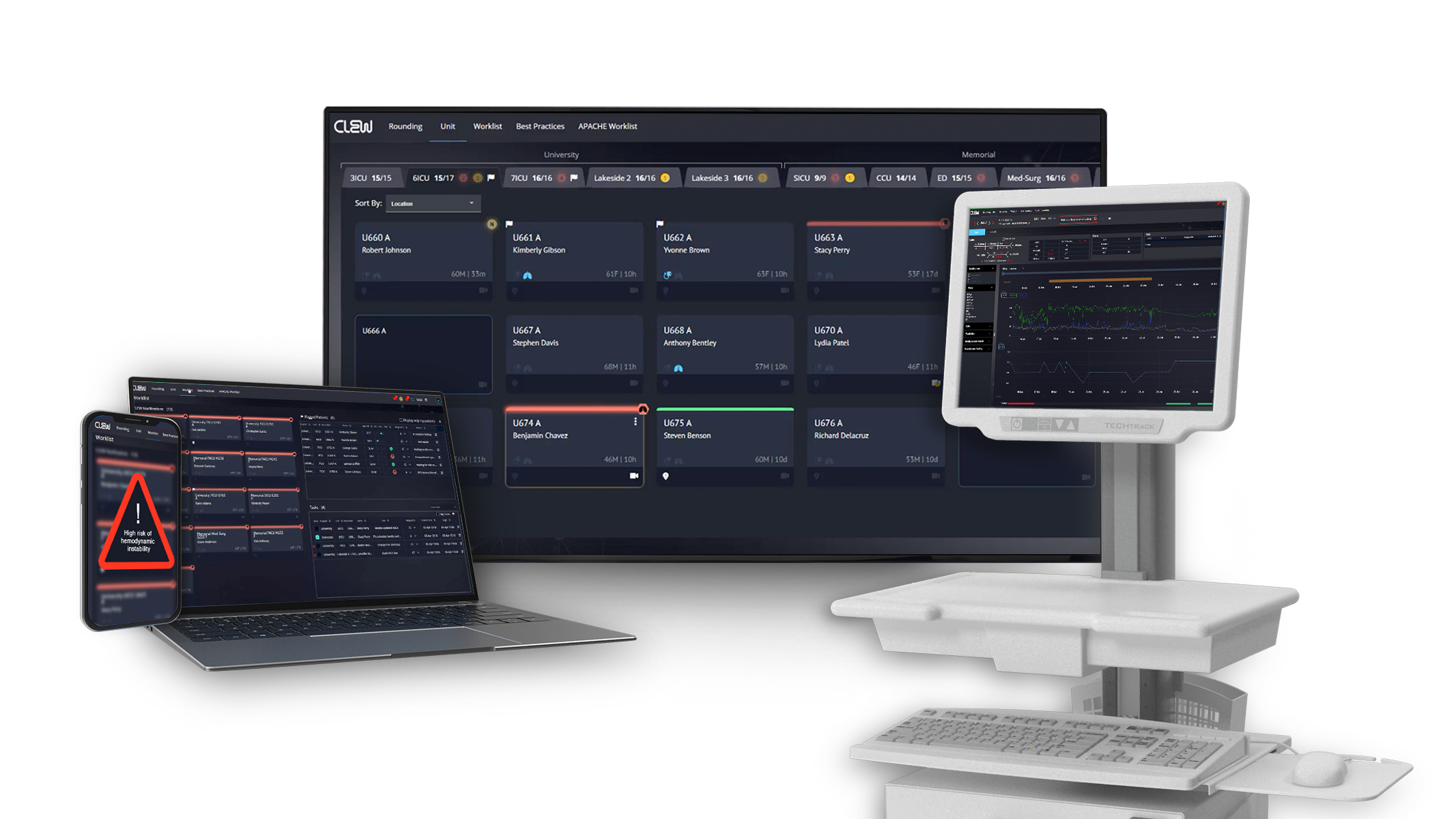
CLEW Medical Scores FDA Clearance for Next-Gen AI-Powered Patient Deterioration Prediction
What You Should Know:
- CLEW Medical, a provider of AI-driven clinical surveillance and predictive analytics, has achieved a breakthrough with FDA 510(k) clearance for its second-generation AI models for predicting patient deterioration.
- The FDA clearance builds upon their pioneering achievement of being the first FDA-cleared Class II medical device for this purpose in 2021.
Rigorous Testing for FDA Approval
Obtaining FDA clearance signifies that CLEW's AI models have
Read More
Philips Receives FDA 510(k) Clearance on Image-Guided Surgical Therapy
What You Should Know:
- Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced its Philips Zenition 30 mobile C-arm has received FDA 510(k) clearance, making image-guided surgical procedures available to more US patients at lower cost.
- By giving surgeons greater flexibility, control, and
Read More
AI Breakthrough for Lung Disease: Thirona’s LungQ Receives FDA Clearance
What You Should Know:
- Thirona, a pioneer in AI-powered lung analysis, received a major boost for its cutting-edge technology LungQ™ with the U.S. Food and Drug Administration (FDA) 510(k) clearance for its latest update, v3.0.0.
- This FDA clearance paves the way for American hospitals to now harness the software's advanced capabilities, marking a significant step forward in lung disease diagnosis and treatment.
LungQ 3.0.0: Precision Navigation for Lung Interventions
This latest
Read More
FemTech: Mosie Baby Awarded FDA Clearance for At-Home Intravaginal Insemination
What You Should Know:
Mosie Baby, a pioneering at-home fertility care company, has secured FDA Class II clearance for its Mosie Baby Kit making it the first and only FDA-cleared over-the-counter kit for use in intravaginal insemination (IVI).The kit was created to support those who are unable to conceive with intercourse or for whom intercourse is not an option. Following its FDA 510k Class II clearance, Mosie Baby looks forward to expanding access to its Mosie Baby Kit which was
Read More
RapidAI Receives FDA Clearance for Rapid SDH (Subdural Hematoma) for Trauma Care
What You Should Know:
- RapidAI has received FDA clearance for Rapid SDH, its AI-powered module for the detection and notification of suspected hemispheric acute and chronic subdural hematoma.
- The need for the RapidAI solution is urgent, with SDH cases in US patients expected to increase by nearly 80% before 2040.
AI Module for Detection of Hemispheric Subdural Hematomas
Rapid SDH leverages AI to help neurocritical care teams identify suspected hemispheric subdural hemorrhage
Read More
Welldoc Awarded FDA 510(k) Clearance for Diabetes Platform
What You Should Know:
Welldoc®, a digital health leader revolutionizing chronic care, today announced the receipt of its 11th 510(k) clearance from the Food and Drug Administration (FDA) for its award-winning diabetes digital health solution, BlueStar®.This clearance immediately follows Welldoc’s 10th 510K clearance announcement earlier this month, solidifying the company’s continued leadership in chronic care innovation.
A Continuous Glucose Monitoring (CGM) Informed Bolus Insulin Dose
Read More
Viz.ai Awarded 510(k) Clearance for AI-Powered Cerebral Aneurysm Detection Solution
What You Should Know:
- Viz.ai, a San Francisco-based provider of AI-powered intelligent care coordination for stroke, has received FDA 510(k) clearance for Viz ANEURYSM (formerly Viz ANX), the first and only AI-powered cerebral aneurysm detection solution designed to facilitate population screening and enhanced care management.
- The new algorithm uses AI to detect suspected cerebral aneurysms, enabling hospital systems
Read More
Biotricity Awarded FDA 510(k) Clearance for Cardiac Monitoring Device
What You Should Know:
- Biotricity, Inc. (NASDAQ:BTCY), a medical diagnostic and consumer healthcare technology company, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Biotres Cardiac Monitoring Device, a three-lead device for ECG and arrhythmia monitoring that is intended for lower-risk patients.
- Most remote cardiac monitors are passive — they don’t record ECG and other cardiac data in real-time — and lack cross-compatibility
Read More
RADLogics Secures FDA Clearance for AI-Powered Chest X-Ray App for Triage & Prioritization
What You Should Know:
- RADLogics today announced that it has secured
510(k) clearance from the FDA for the company’s novel AI-Powered chest X-ray pneumothorax application,
which identifies and prioritizes chest X-ray scans that appear
to contain a pneumothorax, a collapsed lung, for urgent radiologist review.
- RADLogics’ FDA cleared CT and X-ray solutions
– including this application – are
Read More