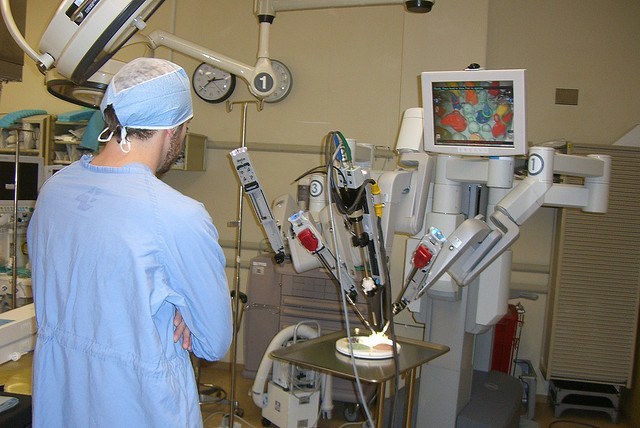
Google is partnering with Ethicon, a medical device company within Johnson & Johnson to create an innovative robotic-assisted surgical platform with the goal of improving health care delivery in the operating room. Under the strategic collaboration, Ethicon will work with Google’s Life Sciences team known as Google X led by Andrew Conrad on advancing surgical robotics to benefit surgeons, patients and health care systems.
Robotic-assisted surgery is a type of minimally invasive surgery that uses technology to give surgeons greater control, access and accuracy during the surgical procedure. For patients, this type of surgery procedure minimizes trauma and scarring, enabling accelerated post-surgical healing. Both companies seek to share capabilities, intellectual property and expertise to develop new robotic tools and capabilities for surgeons and operating room professionals.
“This collaboration with Google is another important step in our commitment to advancing surgical care, and together, we aim to put the best science, technology and surgical know-how in the hands of medical teams around the world,” said Gary Pruden, Worldwide Chairman, Global Surgery Group, Johnson & Johnson.
Approval of the deal would require approval from antitrust regulators at the Federal Trade Commission and Department of Justice. The transaction is expected to be finalized between April and June this year. Financial terms were not disclosed.
Featured image credit: stilldavid via cc
