What You Should Know
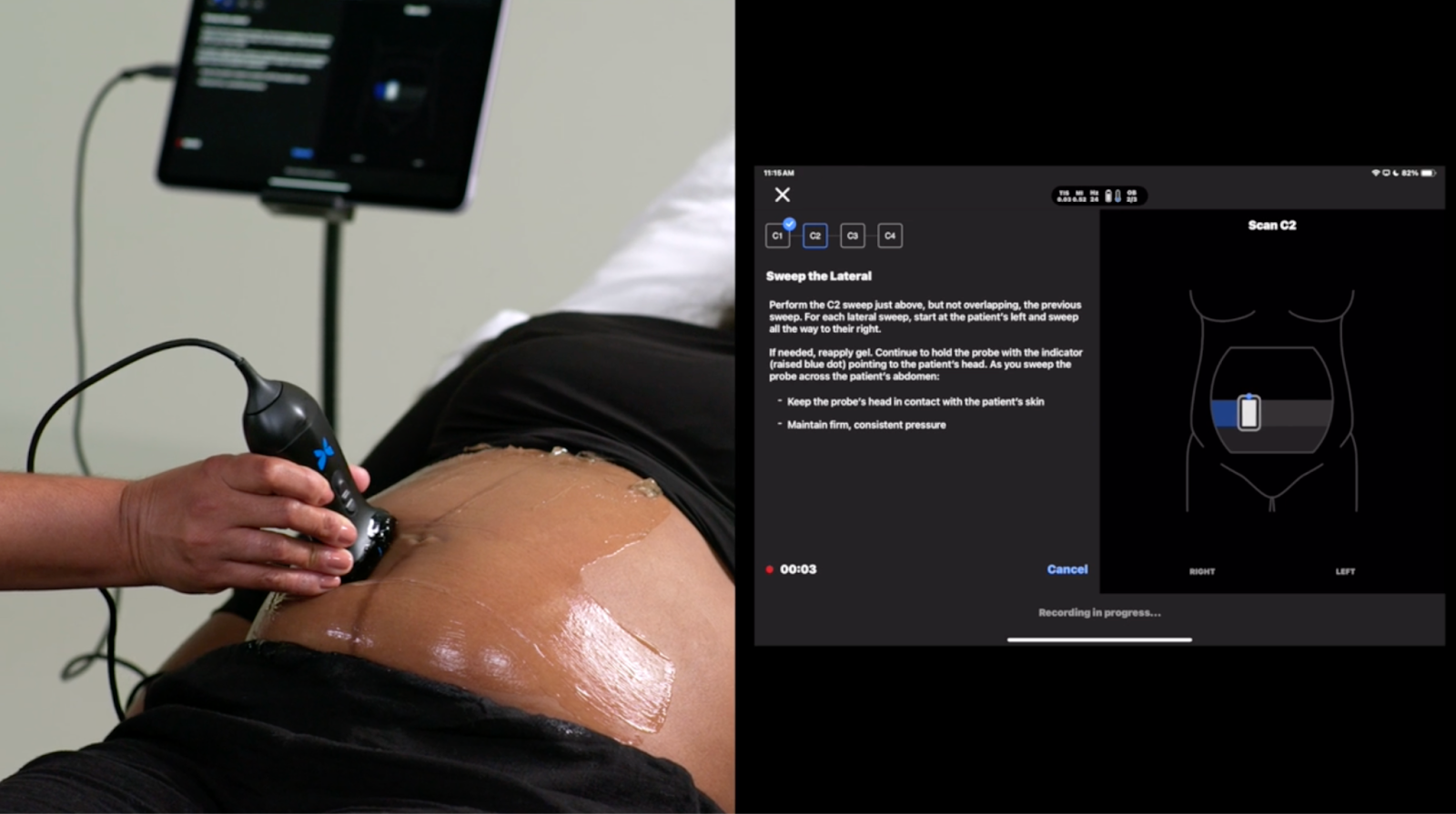
The Milestone: Butterfly Network has received FDA clearance for a fully automated Gestational Age (GA) Tool integrated directly into its handheld, semiconductor-based ultrasound devices. It is the first FDA-cleared "blind-sweep" ultrasound AI tool for estimating gestational age.The Efficacy & Scale: The tool is proven to deliver results equivalent to a trained sonographer for patients between 16 and 37 weeks. Backed by the Gates Foundation, the tool is already
Read More
FDA clearance 510k
GE HealthCare Secures FDA 510(k) Clearance for Photonova Spectra Photon-Counting CT
What You Should Know
The Milestone: GE HealthCare has officially received FDA 510(k) clearance for Photonova Spectra, its highly anticipated photon-counting computed tomography (PCCT) system.The Core Technology: Unlike traditional CT scanners that convert X-rays into visible light before measuring them, PCCT directly counts individual photons and measures their energy. GE is utilizing a proprietary Deep Silicon detector architecture that provides 8-bin energy resolution to virtually eliminate
Read More
HIMSS26: GE HealthCare’s Genesis Radiology Workspace ‘View’ Receives FDA 510(k) Clearance
What You Should Know:
The Clearance: GE HealthCare has officially received FDA 510(k) clearance for View, a fast, zero-footprint diagnostic viewer. This viewer serves as the core anchor of the Genesis Radiology Workspace.The Clinical Bottleneck: Currently, radiologists spend up to 44% of their time on non-interpretive tasks. These tasks include navigating complex interfaces or waiting for images to load, which directly impacts productivity and turnaround times.The Solution: View is engineered
Read More
11 Recent Digital Health FDA Clearances You Need to Know: AI, Wearables, and Robotics Drive Precision Medicine Forward
What You Should Know:
- The U.S. Food and Drug Administration (FDA) has recently issued several key clearances, marking significant advancements across diagnostics, surgery, and remote patient monitoring.
- These FDA clearances reflect a deepening integration of Artificial Intelligence (AI) and advanced technology into clinical workflows, promising greater precision, reduced invasiveness, and expanded access to care.
Diagnostics and Remote Monitoring: AI and Wearables at Scale
A
Read More
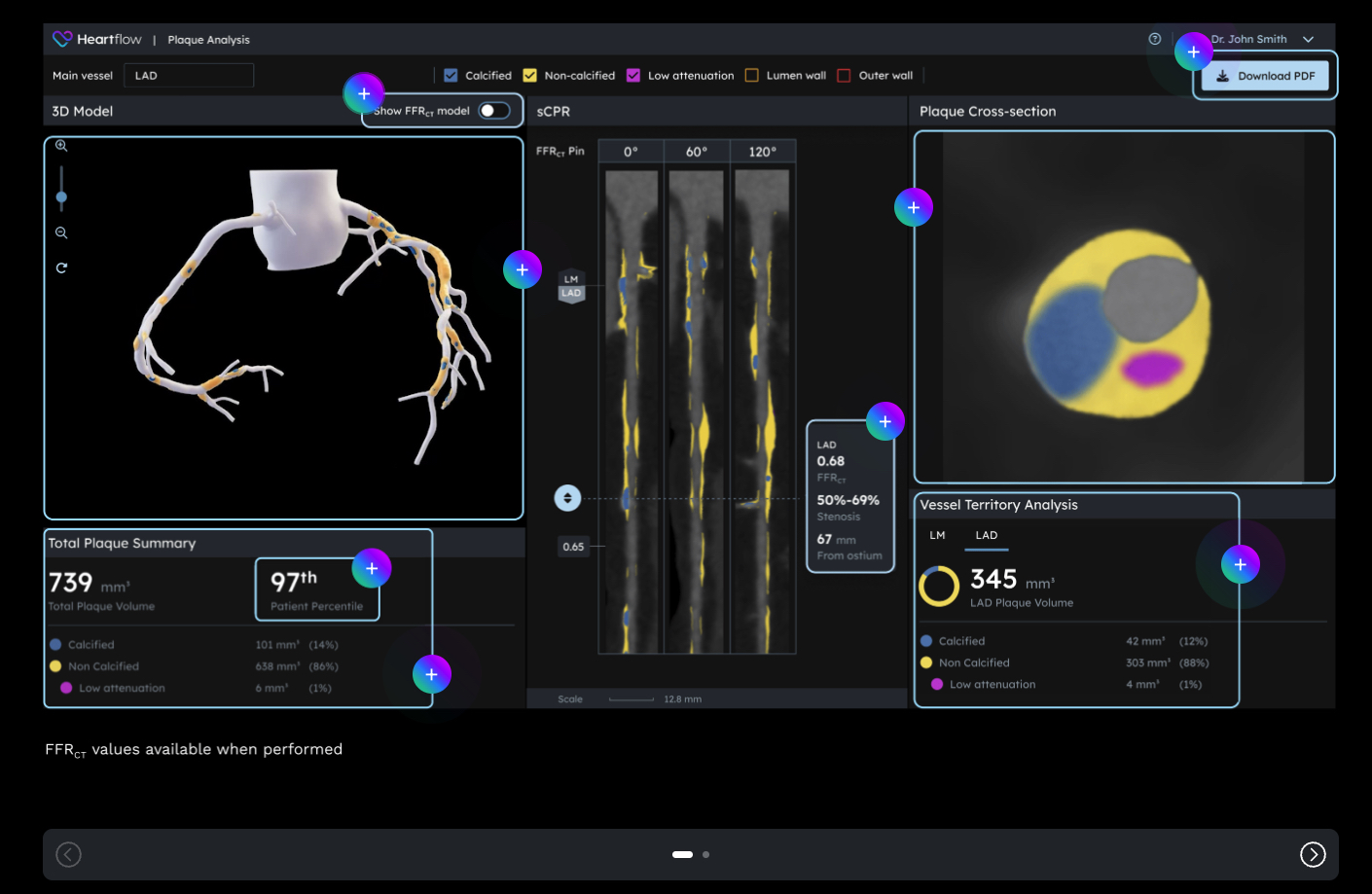
Heartflow Receives FDA Clearance for Next-Gen AI Plaque Analysis, Expands Cigna Coverage
What You Should Know:
- Heartflow, a leader in AI technology for coronary artery disease (CAD), has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Next Gen Heartflow Plaque Analysis algorithm and platform.
- The technology features an updated algorithm, an expanded nomogram, and advanced 3D color-coded visualization of plaque, empowering clinicians with crucial insights for confident care decisions. In addition, Heartflow announced that Cigna will
Read More
PathAI Granted FDA Clearance for AISight Dx for Primary Diagnosis in Clinical Settings
What You Should Know:
- PathAI, a global leader in artificial intelligence (AI) and digital pathology solutions, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its digital pathology image management system, AISight® Dx, for use in primary diagnosis in clinical settings.
- The FDA clearance builds upon the platform's initial 510(k) clearance for AISight Dx(Novo) in 2022, demonstrating PathAI’s ongoing commitment to innovation and
Read More
Withings Sleep Rx Receives FDA Clearance for Home Sleep Apnea Testing
What You Should Know:
- Withings, a global leader in connected health, has announced that its Sleep Rx sleep apnea device has received clearance from the U.S. Food and Drug Administration (FDA).
- Sleep Rx represents a significant advancement in the diagnosis of sleep apnea, offering a more convenient and accurate alternative to traditional in-lab testing.
Addressing the Underdiagnosis of Sleep Apnea
Sleep apnea is a serious condition that affects millions of people
Read More
Philips IntelliSite Pathology Solution Receives 510(k) Clearance
What You Should Know:
- Philips IntelliSite Pathology 5.1 has received 510(k) clearance from the FDA, solidifying its position as the most widely deployed digital pathology solution for primary diagnosis globally.
- With over 300 pathology labs already implementing Philips' solution, and over 20 labs achieving complete digital workflows, the success of this technology is evident.
Investing in the Future of Cancer Care
By empowering collaboration, boosting efficiency, and
Read More
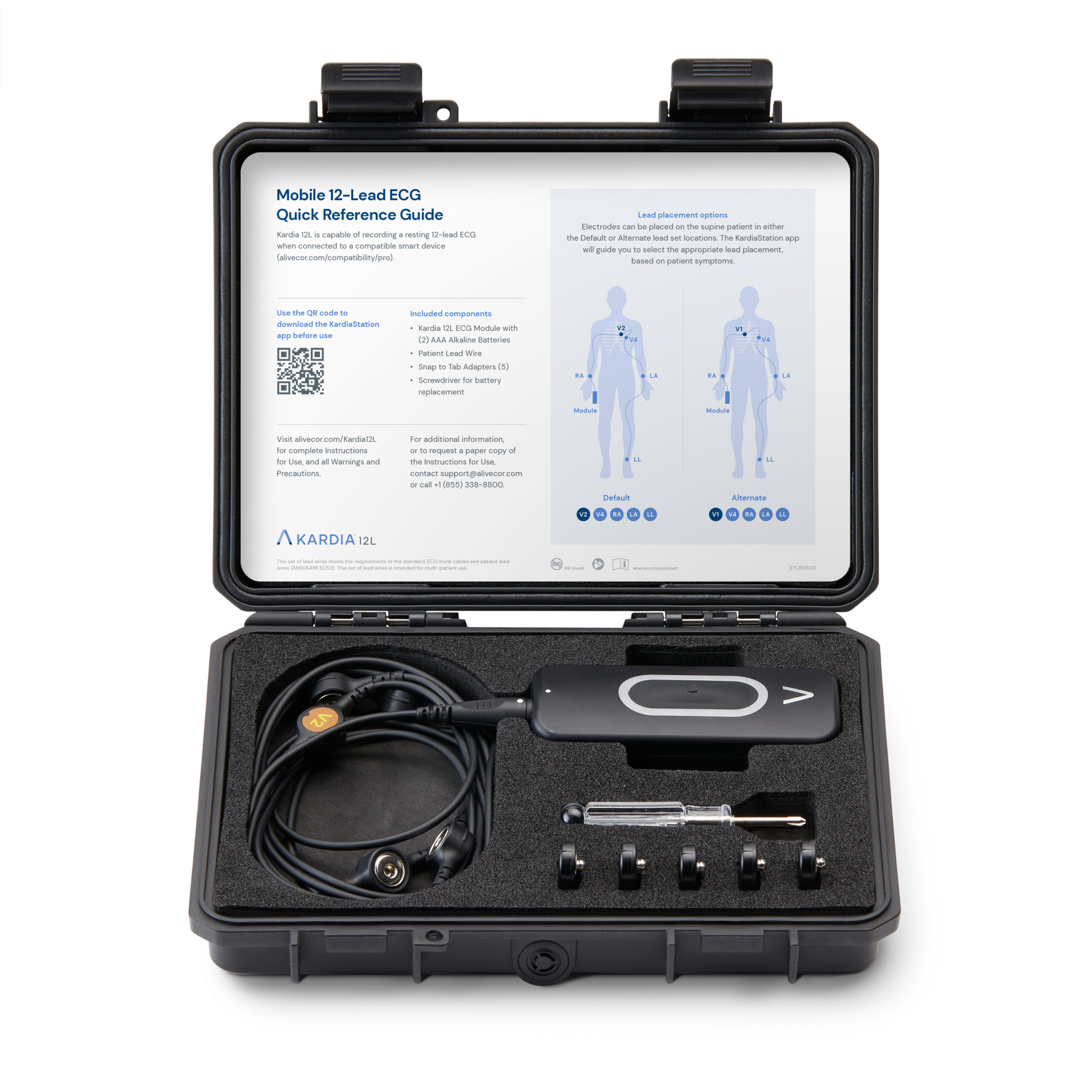
FDA Clears AliveCor’s Heart Attack-Detecting AI Kardia 12L ECG System
What You Should Know:
- AliveCor, a leader in AI-powered cardiology, has received FDA clearance and launched a groundbreaking innovation – the Kardia™ 12L ECG System with KAI™ 12L AI technology.
- AliveCor's Kardia 12L ECG System signifies a major advancement in cardiac diagnostics. By combining cutting-edge AI technology with a portable and user-friendly design, the system empowers healthcare professionals to deliver faster, more accurate, and more accessible cardiac
Read More
WearLinq Acquires AMI Cardiac Monitoring, Raises $6.7M for FDA-Cleared 6-Lead ECG Device
What You Should Know:
- WearLinq, a leader in wearable health monitoring, announced a strategic acquisition of AMI Cardiac Monitoring, LLC, a seasoned player in ambulatory cardiac monitoring.
- The acquisition, coupled with a recent $6.7M funding round, positions WearLinq to significantly expand access to their innovative eWave 6-lead ECG monitor and revolutionize the fight against atrial fibrillation (AFib), which recently received FDA 510(k) clearance.
Tackling the
Read More