What You Should Know:
– CLEW is awarded FDA Emergency Use Authorization (EUA) for its predictive analytics platform in support of COVID-19 patients.
– CLEW is the only device authorized by the FDA to provide early identification of patients who are likely to experience respiratory failure of hemodynamic instability—both significant complications associated with COVID-19.
– CLEW is already implemented in coronavirus units in two Israeli hospitals, and being tested by several US hospitals.
As ICU’s around the world prepare for a potential second COVID-19 wave, the FDA clears the way for use of an early warning/predictive analytics system to proactively manage patient care, ICU teams, and throughput. CLEW announced today that the U.S. Food and Drug Administration (FDA) has issued Emergency Use Authorization (EUA) for the company’s ICU solution. CLEW provides real-time AI analytics platforms designed to help providers make better informed clinical decisions by predicting life-threatening complications across various medical care settings.
From Clinical Data to Life Saving Decisions
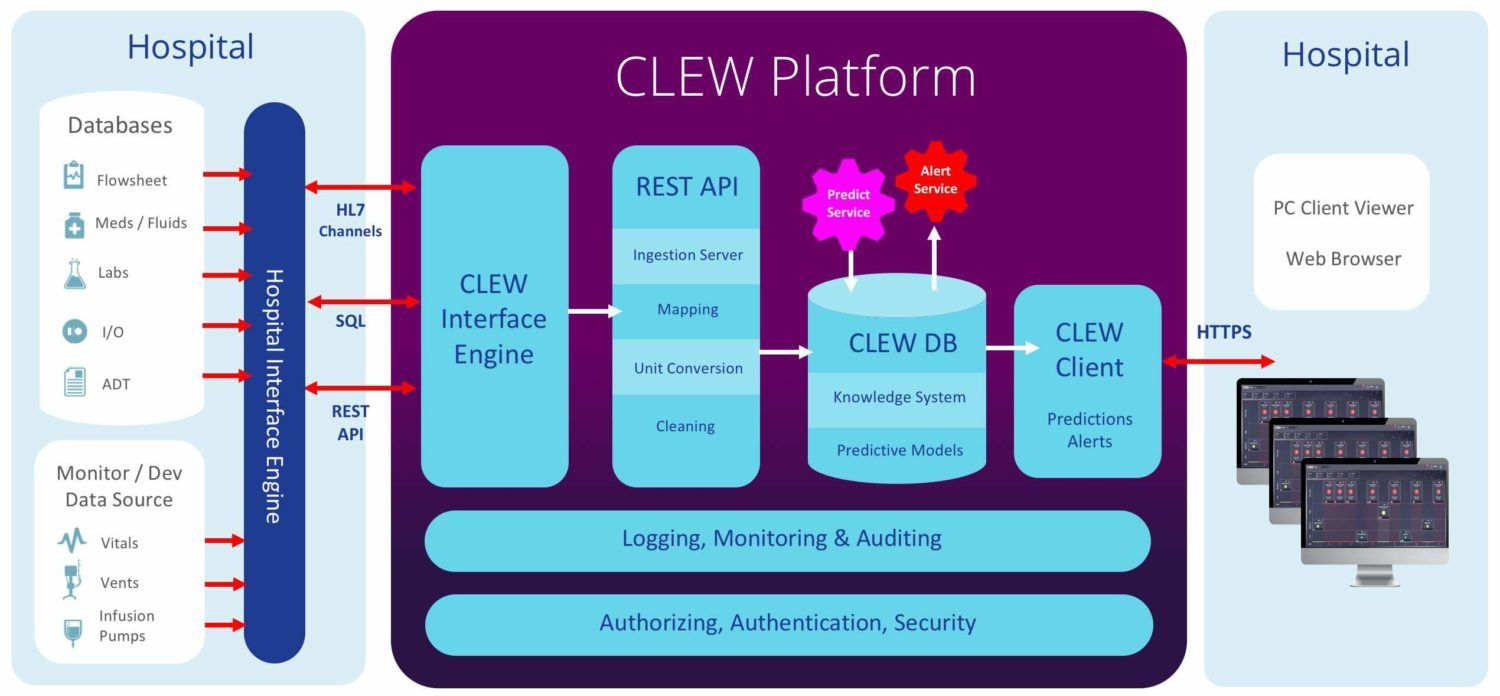
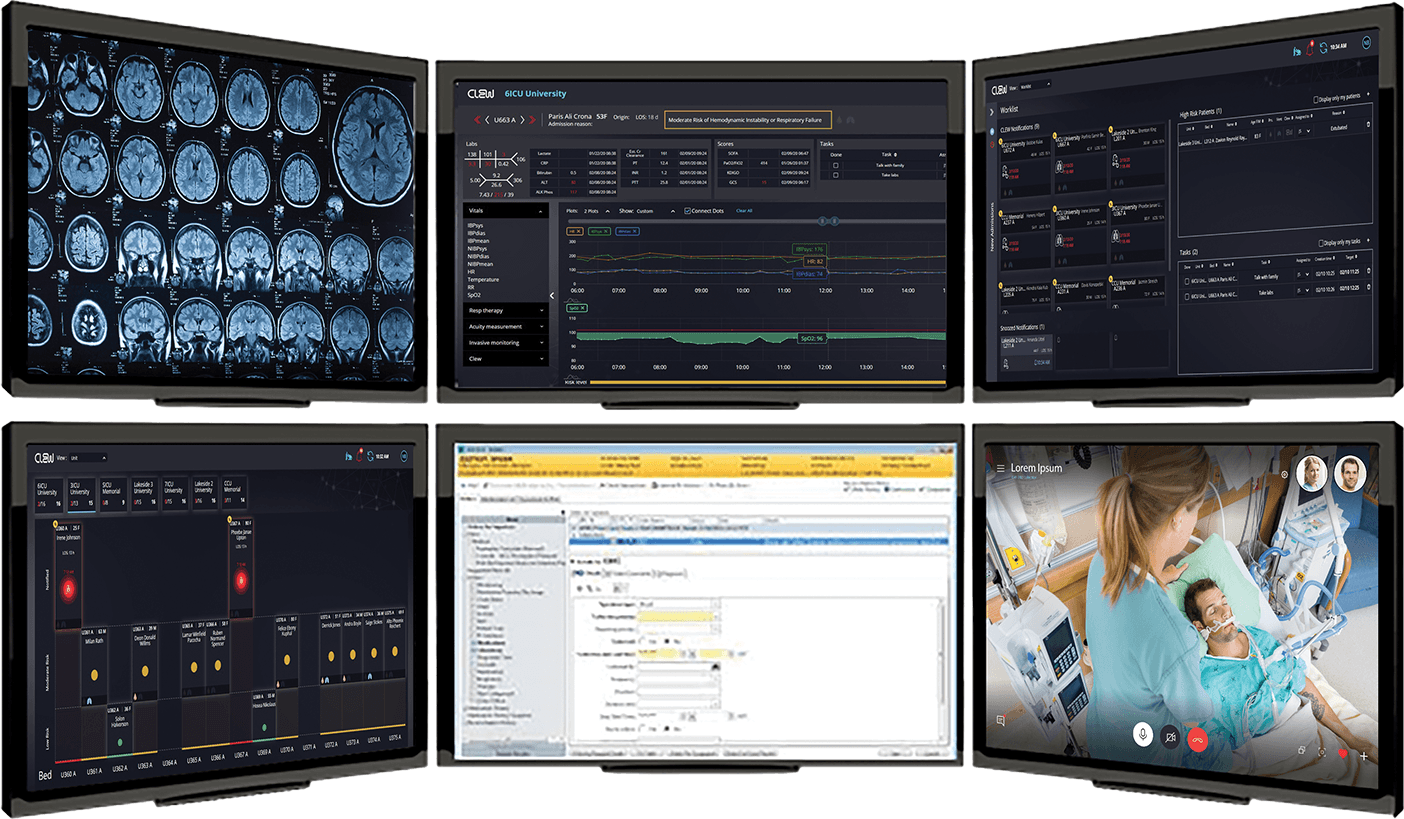
CLEW’s platform harnesses vast amounts of clinical and patient data to deliver highly accurate predictive clinical analytics. CLEW’s analytics engine identifies relationships between real-time physiological data and latent medical conditions, recognizing – in real-time – any changes in the patient’s condition that may indicate the possibility of life-threatening situations. The platform utilizes innovative prediction models derived from Big Data analysis and advanced high-dimensional analytics, to provide hospital management and medical personnel with the preemptive information they require to better manage their resources.
TeleICU Solution by CLEW
With CLEWICU healthcare providers use predictive screening information to help identify patients with an increased likelihood of being diagnosed with respiratory failure or hemodynamic instability which are common complications associated with COVID-19.
– The AI-based algorithms are machine-learning models trained to identify respiratory failure and or hemodynamic instability hours in advance. This allows for additional evaluation and potentially early intervention, planning, resource management.
– CLEW’s AI models were trained on nearly 100,000 patients in the ICUs, and scales to cope with patient volume surges while reducing a caregiver’s exposure risk to infected patients.
– CLEW’s models were developed and intended for both local ICUs and TeleICUs and integrates best practice modules.
– The streamlined at-a-glance web application is designed for near real-time access to patient data and provides tools for both worklist, unit and multiunit views, featuring unit occupancy and patient risk level.
– CLEW-ICU integrates caregiving of local and remote teams enabling workflow and resource decision making.
“Healthcare providers need more than simple analytics. Systems need to integrate into the provider’s workflow, offering ease of use and actionable data. The CLEWICU platform is designed to enable healthcare providers to monitor patient predicted risk levels across all units in real-time allowing for smart decision making about clinical resource allocation, ensuring prompt, proactive and efficient patient care” said Gal Salomon, CLEW CEO.
CLEW is already implemented in coronavirus units in two Israeli hospitals, and being tested by several US hospitals.
