Today, Philips announced it has received regulatory clearance from the FDA (via De Novo classification) for its IntelliSite Pathology Solution, marking a significant leap forward for the pathology services industry in the U.S. De novo classification is the regulatory pathway for marketing clearance for novel, low- to moderate-risk medical devices that are the first of their kind
As the first and only digital pathology system to be approved for primary diagnostic use in the country, today’s news has implications not only for improving laboratory workflow and efficiencies, but also for improving the quality and accuracy of cancer diagnostics through computational pathology—an approach to rendering disease diagnoses that incorporates multiple sources of data and uses mathematical models to generate clinically actionable inferences.
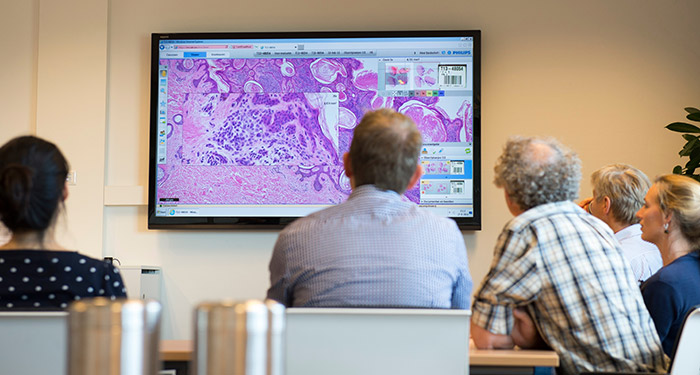
Philips IntelliSite Pathology Solution is an automated digital pathology image creation, viewing and management system comprised of an ultra-fast pathology slide scanner, an image management system and a display. This solution is complemented by advanced software tools to manage the scanning, storage, presentation, reviewing, and sharing of information. By supporting the transition to digital workflows, Philips seeks to help pathology laboratories simplify access to histopathology information and help staff work more efficiently.
The FDA evaluated data from a clinical study of approximately 2,000 surgical pathology cases. This was one of the largest studies ever conducted to directly compare the use of digital pathology to optical microscopes. Sixteen pathologists at four clinical study sites – Cleveland Clinic, University of Virginia, Miraca Life Sciences and Advanced Pathology Associates – conducted approximately 16,000 reads across 2,000 cases. FDA’s news release ‘FDA allows marketing of first whole slide imaging system for digital pathology’ can be found here.
Pathology services are under pressure to meet the increasingly complex demands of cancer care. As the number of cancer cases continue to grow, the complexity and number of tests have simultaneously increased to support improved patient care. Pathologists must work as efficiently and effectively as possible to analyze the complex information that influences the design of personalized treatments. Outside the U.S., where CE-IVD mark has allowed primary diagnosis since 2014, digital pathology is recognized for increased efficiencies in the logistics and consultation needs in primary diagnosis of histopathology images.
It is an exciting time in pathology as paradigm shifts are being witnessed in cancer care with precision medicine and personalized treatments increasing by the day – pathologists are absolutely central to this dream of personalized medicine.
“The clearance for Philips IntelliSite Pathology Solution marks a major milestone for innovation in pathology services in the U.S.,” said Russ Granzow, General Manager of Philips Digital Pathology Solutions. “Not only will it promote increased efficiencies and collaboration between pathologists, but it also opens a complete new dimension towards computational pathology which aims to increase accuracies and ultimately enhance patient care.”
