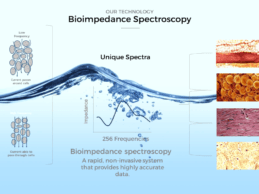
ImpediMed Ltd, a Australia-based medical device startup has signed a three-year joint development agreement with Mayo Clinic to explore uses for body fluid status monitoring technology. ImpediMed employs bioimpedance spectroscopy (BIS) technologies for use in the non-invasive clinical assessment and monitoring of fluid status in patients. ImpediMed has the first medical device with FDA clearance in the U.S. to aid healthcare professionals to clinically assess secondary unilateral lymphedema of
Read More
- Skip to main content
- Skip to secondary menu
- Skip to primary sidebar
- Skip to secondary sidebar
- Skip to footer