What You Should Know
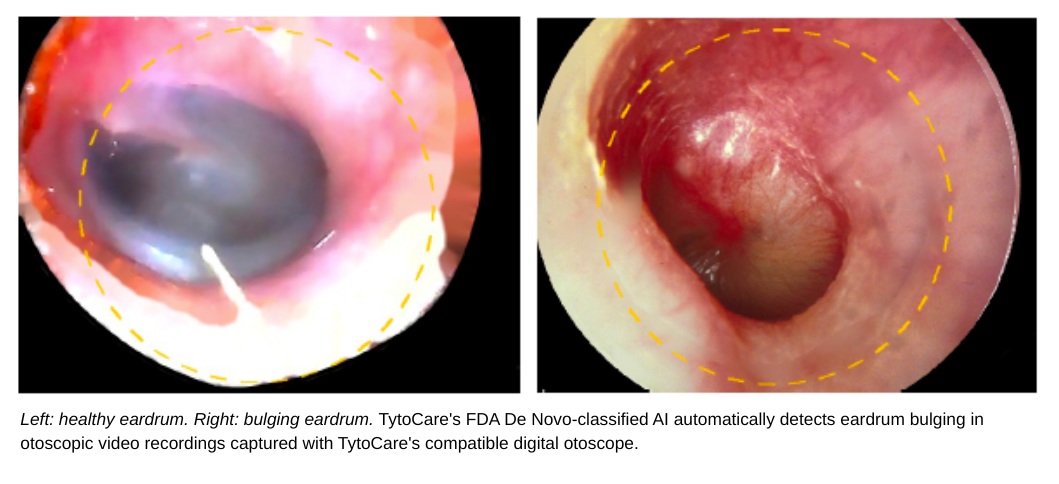
TytoCare has received FDA De Novo classification for its Tyto Insights™ for ENT Suite, creating a first-of-its-kind regulatory category for "ear, nose, and throat image analyzers".The AI-powered tool automatically analyzes otoscopy video to detect eardrum bulging, a primary indicator of Acute Otitis Media and other common pediatric conditions.Designed for patients aged six months and older, the system uses a proprietary database of 1.6 million ear images to provide
Read More
FDA Clearance
Butterfly Network Receives FDA Clearance for AI-Powered Gestational Age Ultrasound Tool
What You Should Know
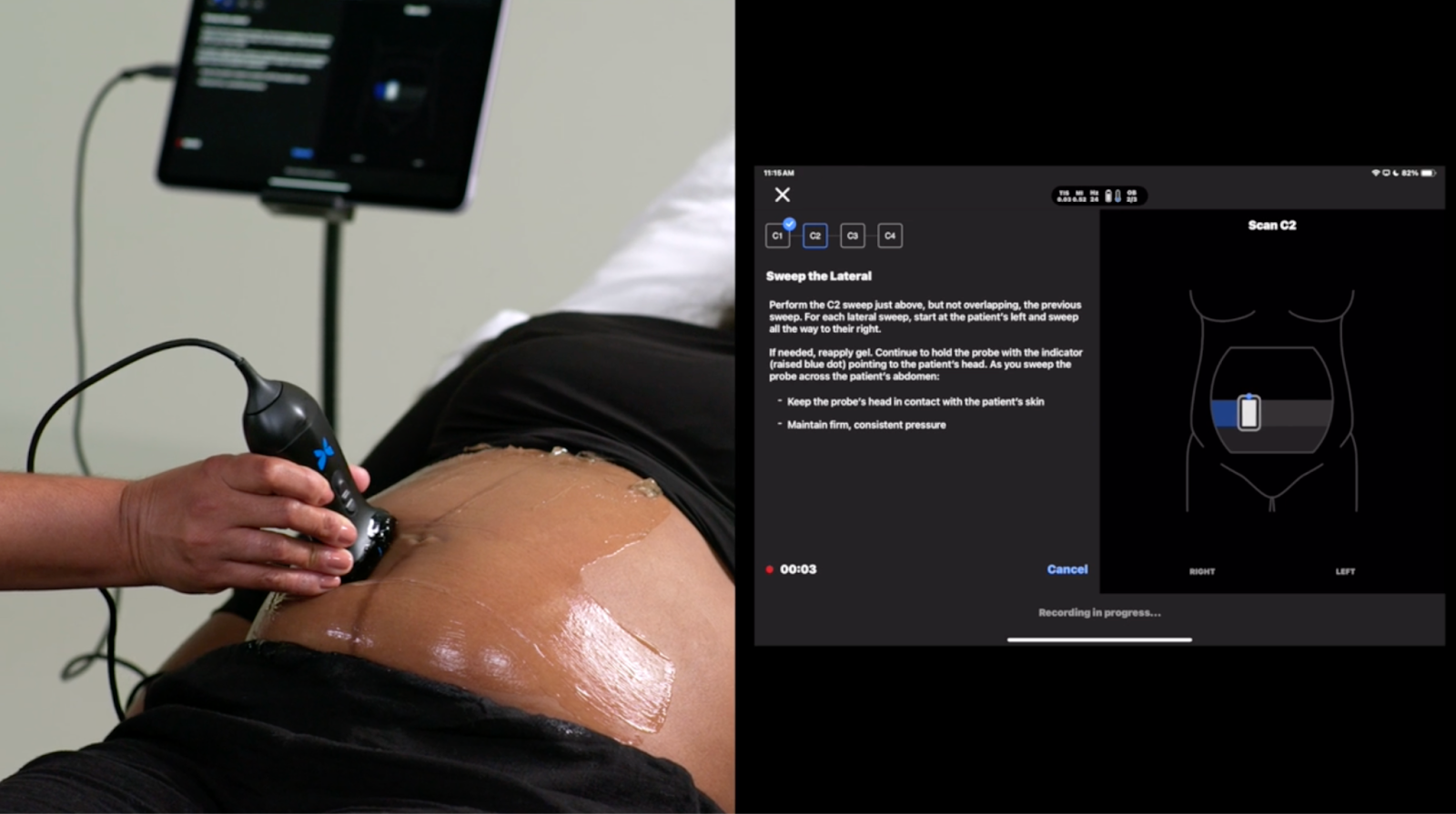
The Milestone: Butterfly Network has received FDA clearance for a fully automated Gestational Age (GA) Tool integrated directly into its handheld, semiconductor-based ultrasound devices. It is the first FDA-cleared "blind-sweep" ultrasound AI tool for estimating gestational age.The Efficacy & Scale: The tool is proven to deliver results equivalent to a trained sonographer for patients between 16 and 37 weeks. Backed by the Gates Foundation, the tool is already
Read More
GE HealthCare Secures FDA 510(k) Clearance for Photonova Spectra Photon-Counting CT
What You Should Know
The Milestone: GE HealthCare has officially received FDA 510(k) clearance for Photonova Spectra, its highly anticipated photon-counting computed tomography (PCCT) system.The Core Technology: Unlike traditional CT scanners that convert X-rays into visible light before measuring them, PCCT directly counts individual photons and measures their energy. GE is utilizing a proprietary Deep Silicon detector architecture that provides 8-bin energy resolution to virtually eliminate
Read More
HIMSS26: GE HealthCare’s Genesis Radiology Workspace ‘View’ Receives FDA 510(k) Clearance
What You Should Know:
The Clearance: GE HealthCare has officially received FDA 510(k) clearance for View, a fast, zero-footprint diagnostic viewer. This viewer serves as the core anchor of the Genesis Radiology Workspace.The Clinical Bottleneck: Currently, radiologists spend up to 44% of their time on non-interpretive tasks. These tasks include navigating complex interfaces or waiting for images to load, which directly impacts productivity and turnaround times.The Solution: View is engineered
Read More
GE HealthCare Receives FDA Clearance for Allia Moveo Interventional Imaging System
What You Should Know
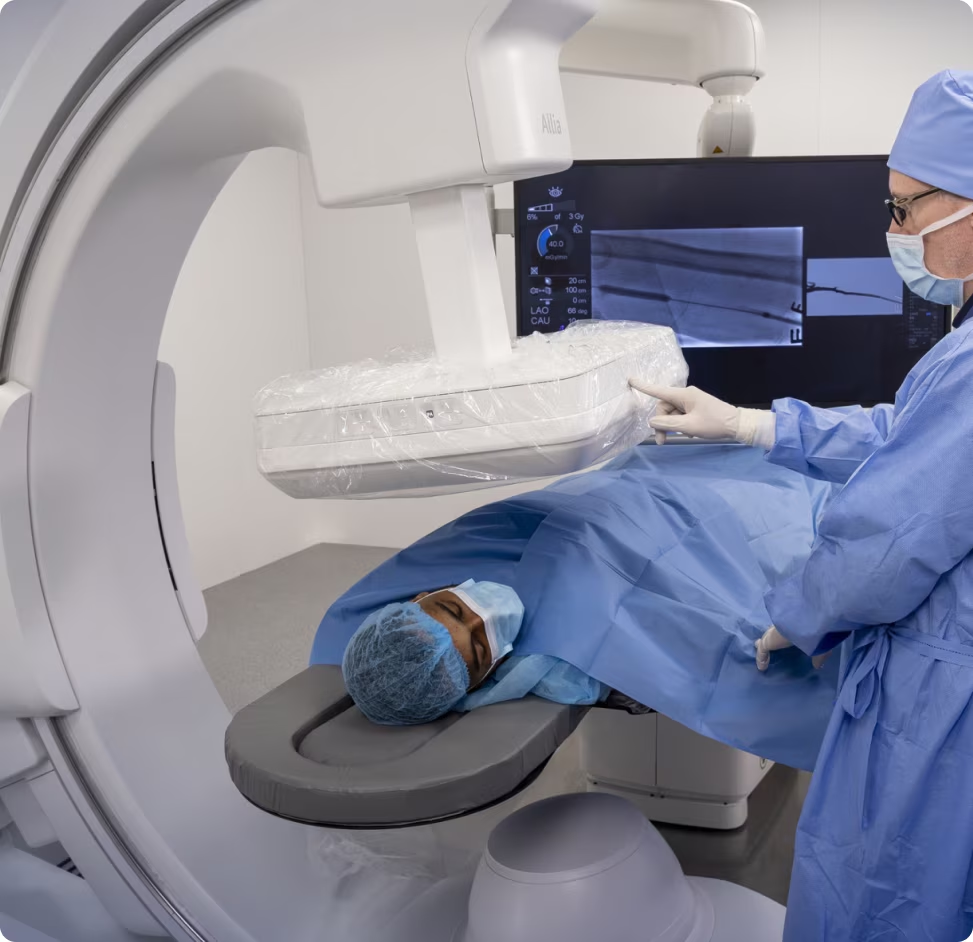
The Launch: GE HealthCare has received FDA 510(k) clearance and CE Marking for Allia™ Moveo, a new mobile C-arm system designed for cardiovascular and interventional procedures.The Design: The system addresses the physical constraints of the operating room. It is compact and cable-free, featuring a "wide-bore" design that accommodates patients of any size while allowing effortless table panning.The AI: The platform integrates CleaRecon DL, an AI-driven tool that removes
Read More
Proprio Secures 4th FDA Clearance for AI-Powered ‘Picasso’ Spine Guidance
What You Should Know:
- Proprio has secured FDA clearance for Picasso, its fourth cleared capability within the Paradigm AI surgical platform.
- The feature enables "trace-based" optical registration, allowing spine surgeons to perform real-time, radiation-free 3D measurements of spinal alignment during surgery—addressing a critical gap where surgeons previously relied on static 2D X-rays or postoperative confirmation.
Radiation-Free "Digital Twins"
Legacy navigation
Read More
AliveCor Receives FDA Clearance for 39 Determinations on Kardia 12L AI
What You Should Know
- AliveCor, the global leader in AI-powered cardiology, has received U.S. FDA clearance for the next generation of its KAI 12L AI. This update expands the diagnostic breadth of the Kardia 12L ECG System to detect five additional cardiac determinations, bringing the platform’s total to 39 cleared determinations.
- The handheld, 0.3-pound device can now identify specific rhythm modifiers and axis-related morphologies, providing clinicians with unprecedented diagnostic
Read More
AccurKardia Secures Second FDA Clearance for AccurECG 2.0 to Tackle Cardiac Backlogs
What You Should Know
- AccurKardia has announced its second FDA 510(k) clearance for the AccurECG™ Analysis System (v2.0). This enterprise-grade, cloud-based platform is designed to ingest data from any ECG hardware—including patches and Holters—and deliver fully automated, near real-time interpretation for 13 rhythm classifications with 99% accuracy.
- As cardiac data volumes surge due to the proliferation of wearables, AccurECG 2.0 allows hospitals and independent diagnostic testing
Read More
RapidAI Secures FDA Clearance for Five New Deep Clinical AI Modules, Expanding Enterprise Imaging Platform
What You Should Know:
- RapidAI, a pioneer in deep clinical AI, announced U.S. FDA clearance for five new imaging modules—Rapid DeltaFuse™, Rapid LMVO, Rapid MLS, Rapid OH, and Rapid Aortic for measurement.
- The expansion reinforces the Rapid Enterprise™ Platform’s focus on bringing deep clinical intelligence and seamless workflow integration across the entire patient journey.
Deep Clinical AI: Shifting Beyond Triage to Full Patient Management
RapidAI’s latest FDA clearances
Read More
Ceribell Receives FDA Clearance for Clarity® AI, Becoming First Point-of-Care EEG for All Ages
What You Should Know:
- Ceribell, Inc. today announced that the U.S. FDA has granted 510(k) clearance for its next-generation Clarity® algorithm.
- The FDA clearance makes the Ceribell System the first and only AI-powered point-of-care electroencephalography (EEG) technology available to detect electrographic seizures in all patients, from pre-term neonates through adults.
Closing a Critical Unmet Need in Neonatal Care
The FDA clearance for the Clarity® algorithm to
Read More