Remote patient monitoring yielded a savings of $8,375 per patient annually, which, for a typical one-million-member health plan could translate into about $1.1 million saved each year. The results stem from a year-long remote patient monitoring pilot from Geneia,an analytics and technology leader in transforming healthcare delivery.
@Home Pilot Study Overview
The @Home pilot study compared the experience of Capital BlueCross members diagnosed with heart failure (HF), who enrolled and actively participated in the @Home remote monitoring program, to a control group of non-monitored members with statistically similar pre-program risk scores. The study evaluated clinical, utilization and cost outcomes, and patient experience
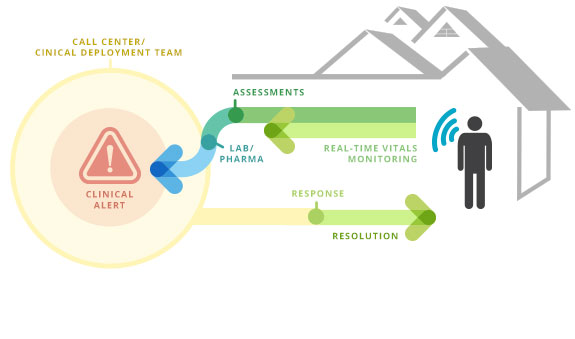
Geneia’s @Home remote patient monitoring program, powered by Medtronic’s ZephyrLIFETM technology, is a 24x7x365 solution that helps chronically ill patients and their providers better understand, monitor and manage their health.
The program combines predictive analytics, state-of-the-art technology and in-home clinical support to identify chronically ill patients most likely to participate in and benefit from the program. It then supports them through personalized care planning to improve health outcomes.
“The @Home remote patient monitoring program is an important and successful component of a robust and comprehensive case management strategy at Capital BlueCross,” said Chambers. “Not only is chronic disease expensive, difficult to manage and a drain on our healthcare system, but it also takes a toll on patients and families, decreasing their quality of life and often leading to an accelerated transition out of the home and into facility-based care.”
“The results of the @Home remote monitoring pilot program are promising,” continued Chambers. ”We are confident that more of our seniors struggling with chronic disease will benefit from this technology and, together with their provider and the case management team at Geneia, learn to better understand and manage their chronic conditions and continue to live full and independent lives in their own homes.”
Key findings from the @Home study include:
– Slowed disease progression, as measured by patient risk scores. The risk score stabilized: 2 percent increase in the pilot group compared to 31 percent increase in the control group.
– Reduction in hospitalizations:
– Net 45 percent reduction in acute hospital admissions (66 percent reduction in the pilot group and a 21 percent reduction in the control group).
– Net 34 percent reduction in HF acute hospital admissions (90 percent reduction in the pilot group and 56 percent reduction in the control group). Note: HF acute hospital admissions are a subset of acute hospital admissions.
– The reduction in hospital admissions means an average per-participant, per-year savings of $8,375 in monitored HF patients. As an example, taking into consideration eligibility and expected participation rates and subtracting program costs, a typical health plan with one million members, could expect to save about $1.1 million per year.
Improved patient experience:
– @Home study participants reported an overall satisfaction rate of 96 percent.
– 96 percent said “…my @Home nurse helped me to better understand my health condition(s) and identify and manage my healthcare needs.”
– 97 percent said “…my @Home nurse helped me understand when I should talk to my doctor or healthcare provider about symptoms or changes in my condition.”
– One unexpected result was an increase of 7 percent in ED visits in the pilot group. However, this is consistent with other research that reported monitoring HF patients was associated with a reduction in planned hospital visits and lower monetary costs, despite a modest increase in unplanned hospital and ED visits (Klersey, 2016).
@Home Study Pilot Study Background/Methodology
Participants in the study and control groups had:
– A diagnosis of heart failure
– At least one inpatient admission or two or more emergency department (ED) visits in the 12 months prior to being recruited for participation in the program
– A statistically similar risk score, as measured by a retrospective claims- and demographic-based risk score
– 12 months of pre-program claims data and three to 12 months of post-program claims data
Eligible study participants included in the @Home pilot study received 91 to 180 days of monitoring with a minimum 50 percent compliance rate between January 2015 and February 2016. The study evaluated:
– Clinical outcomes and disease progression, as measured through a change in risk score
– Utilization outcomes, as measured through acute hospital admissions and HF-specific inpatient hospital admissions
– Financial outcomes
