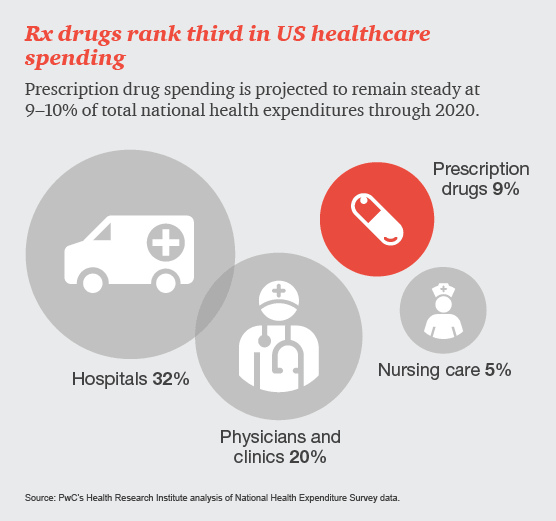
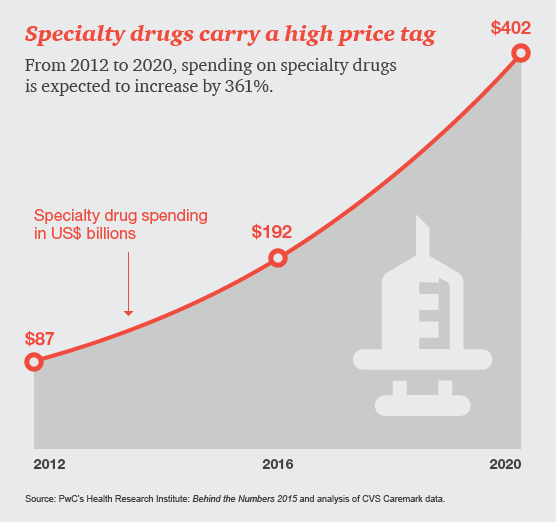
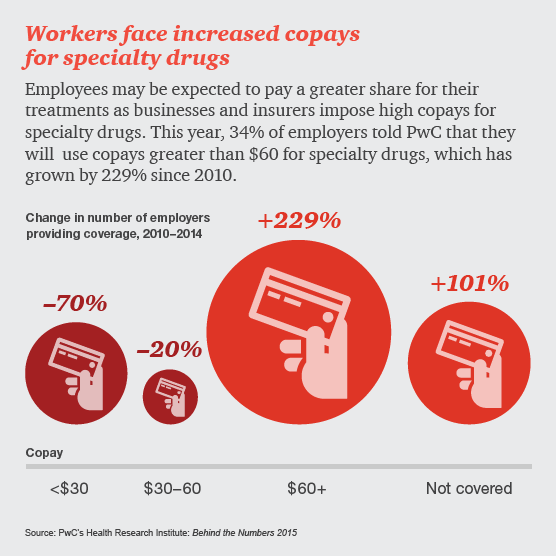
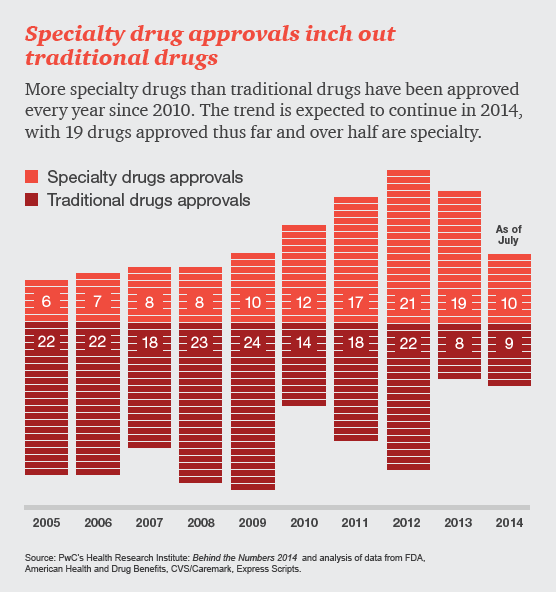
Drug and device manufacturers face mounting pressures from an evolving health industry that places a premium on speedy access to breakthrough, cost-effective and safe medical products.
The $347 billion-a-year industry answers to a new class of consumers who shoulder more of their treatment costs and who demand a greater role in product development. Companies working to invent novel therapies and win regulatory approval face the added pressure of demonstrating the value those products bring.
These trends force the pharmaceutical and life sciences industry to reexamine interactions with its most important stakeholders: the Food and Drug Administration (FDA) and consumers.
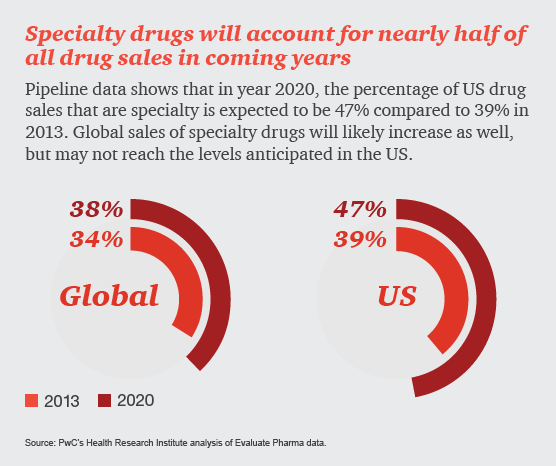
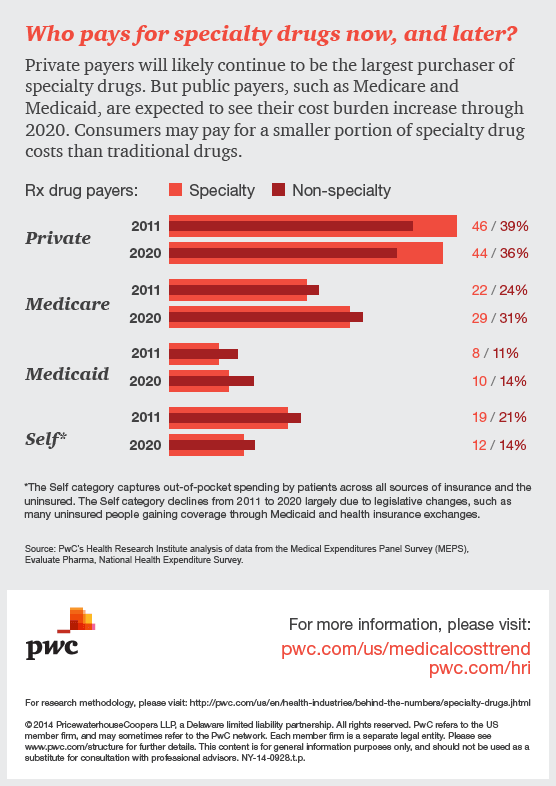
Specialty drugs, used to treat complex illnesses such as cancer and hepatitis C, are in the spotlight, sparking discussion around how society will pay for high-cost therapies in the future. Last year, 70% of all medication approvals by the Food and Drug Administration (FDA) were for specialty drugs. Many more are in the pipeline. In this infographic, HRI explores how specialty drugs are impacting the US healthcare system now and in years to come.