What You Should Know
- The Adoption Curve: KLAS Digital Pathology 2026 Report reveals the US digital pathology market is rapidly growing due to recent FDA clearances and progress toward specific reimbursement. However, adoption remains in the early stages, with fewer than 15% of US healthcare organizations having selected a digital pathology vendor.
- The IMS Leaders: When it comes to Image Management Systems (IMS), pathology-specific vendors like Proscia, PathAI, and LUMEA are currently outperforming traditional radiology vendors in early customer satisfaction. Customers praise their purpose-built platforms and strategic guidance.
- The Hardware Heavyweight: Leica Biosystems is the undisputed market leader for clinical scanners in the US. Customers consistently highlight its high-quality scans, efficient slide loading, and high day-to-day reliability. Conversely, Roche scanner customers report significant dissatisfaction due to reliability issues and low throughput.
- The AI Focus: Hospitals are primarily evaluating AI for clinical use, particularly for breast and prostate cancer algorithms. A major requirement for these AI tools is that they must integrate directly into the IMS workflow rather than functioning as a separate system.
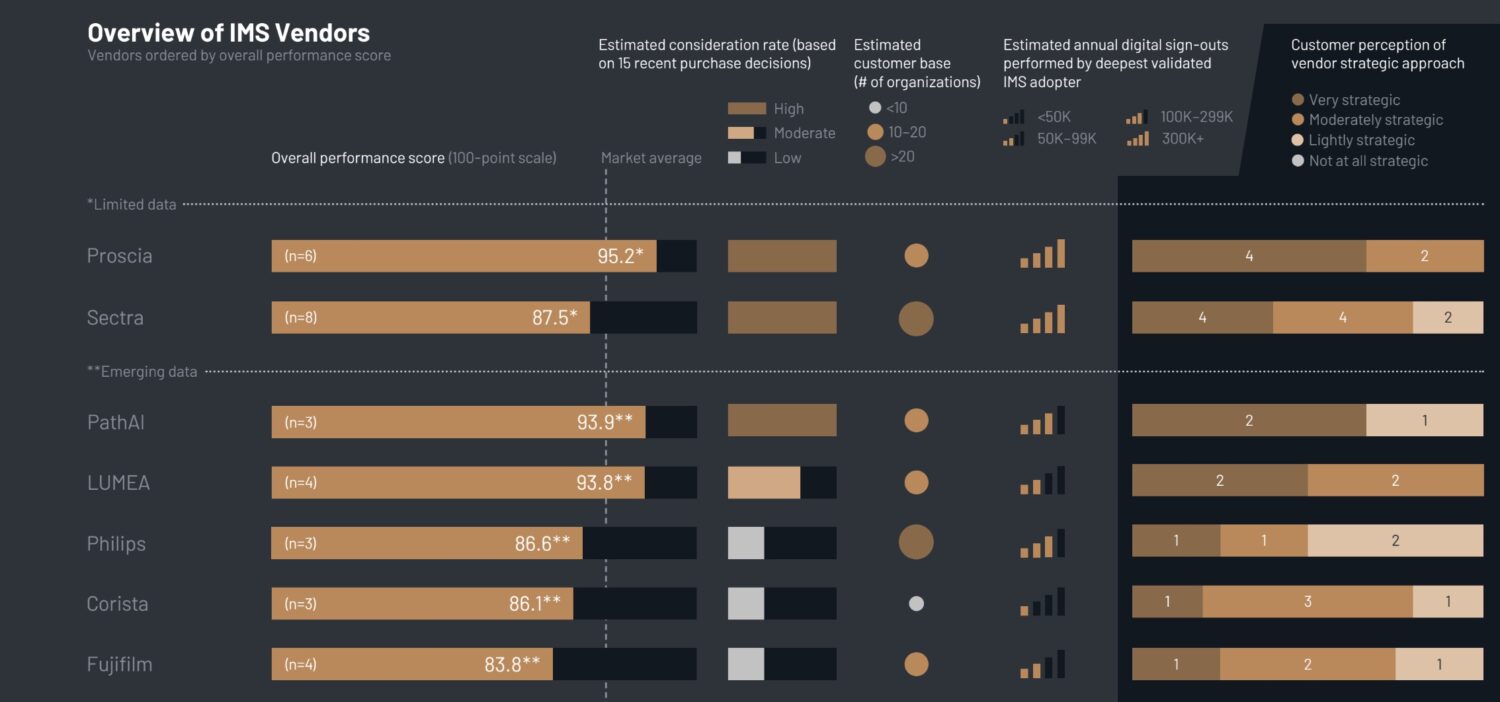
The IMS Battle: Specialists vs. Generalists
The Image Management System (IMS) is the software backbone of digital pathology. The KLAS data reveals a clear divide in this market between two types of vendors: early entrants that expanded into pathology from a broader radiology background (like Sectra, Philips, and Fujifilm), and newer vendors built specifically for pathology.
Right now, the specialists are winning the satisfaction battle.
- Proscia is the most frequently considered IMS vendor. Customers cite their highly collaborative, education-focused approach as a key differentiator.
- PathAI is receiving positive initial feedback centered around proactive communication, hands-on integration efforts, and visible executive engagement.
- LUMEA customers praise the solution’s ease of use and the vendor’s hands-on, strategic engagement to adapt workflows to customer-specific needs.
While traditional imaging vendors like Sectra continue to grow quickly thanks to their established radiology PACS relationships , some customers report that post-go-live engagement feels limited. As organizations mature, they are demanding more proactive guidance, especially around AI interoperability.
The Hardware Reality: Scanning at Scale
Software is useless without a digitized image. In the scanner hardware market, Leica Biosystems is dominating. As one of the first to receive FDA clearance, their Aperio GT 450 DX scanner is the most widely adopted for clinical use in the US. Customers consistently highlight the scanner’s high-quality scans, efficient slide loading, strong throughput, and day-to-day reliability as massive productivity drivers.
On the other end of the spectrum, Roche customers are expressing significant dissatisfaction. Echoing global research trends, US respondents report major challenges scaling Roche scanners for primary diagnosis, citing reliability issues and low throughput that disrupt high-volume workflows.
The AI Integration Imperative
The ultimate promise of digital pathology isn’t just viewing a slide on a monitor; it is unleashing Artificial Intelligence on the pixels. Early indicators point to strong and growing interest in AI. Organizations are no longer just looking at research; they are evaluating AI for direct clinical use. Breast and prostate cancer algorithms have emerged as the most common focus areas. Specifically, interest is strongest in breast cancer applications that support biomarker assessment, such as Ki-67, HER2, estrogen receptor (ER), and progesterone receptor (PR).
The top AI developers currently being considered include Ibex, Visiopharm, Paige.ai, and PathAI. However, there is a hard operational mandate from the buyers: integration is non-negotiable. Respondents consistently emphasize that AI must integrate directly into the IMS workflow. Hospitals expect AI to function as a seamless part of routine clinical operations, rather than forcing pathologists to log into a separate, disconnected system.
For more information about this report, visit https://klasresearch.com/report/digital-pathology-2026-early-adoption-and-technology-performance-in-an-emerging-us-market/3711
