What You Should Know:
- Aetion, a provider of real-world evidence (RWE) technology and analytics, today announced that the FDA has extended Aetion’s contract to evaluate real-world data (RWD) involving COVID-19 medical countermeasures.
- Aetion will be expanding its work to evaluate data standardization in the context of COVID-19. Aetion has been collaborating with FDA since May 2020, providing validated software platform capabilities and RWD/E expertise to contribute to rapid evidence
Read More
Aetion
Aetion and Cognito Team Up for Predictive Modeling in Alzheimer’s
What You Should Know:
- Aetion, real-world evidence (RWE) tech and analytics provider, and Cognito Therapeutics, a pioneer in neuromodulation therapeutics for neurodegenerative diseases like Alzheimer’s, are partnering to develop an AI-driven predictive model to help payers and providers identify patients with rapidly progressing mild cognitive impairment (MCI) and Alzheimer’s who may benefit from earlier treatment.
Impact of MCI & Alzheimer’s Disease
There are approximately 13.5
Read More
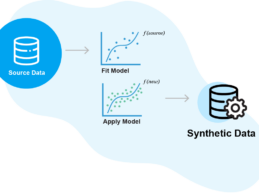
Aetion Acquires Synthetic Data Platform Replica Analytics
What You Should Know:
- Aetion, a real-world evidence (RWE) tech platform, has just announced its acquisition of Replica Analytics, a pioneer in using ML to generate synthetic data. Financial details of the acquisition were not disclosed.
- This acquisition allows Aetion to solve for one of the toughest problems in RWE: access to fit-for-purpose data. Synthetic data acts as a “digital clone” of real-world data by maintaining the source data’s statistical properties, while also protecting
Read More
FDA Selects Aetion Evidence Platform to Assess In-Patient COVID-19 Treatments
What You Should Know:
- Aetion, a NYC-based leading real-world evidence (RWE) company, announced that the U.S. Food and Drug Administration (FDA) has contracted with them to use their Aetion Evidence Platform® to rapidly assess in-patient COVID-19 treatments and advance regulatory science to help determine when, where and how to use RWD.
- This project is designed to demonstrate how using a platform-based approach
Read More
Syneos Health, Aetion Partner to Offer RWE Solutions to Advance Drug Development and Commercialization
What You Should Know:
- Aetion, a leader in real-world evidence analysis, and Syneos Health® (Nasdaq:SYNH), one of the largest CROs, announced a partnership to provide regulatory-grade data and analytics-driven solutions to advance drug development and improve patient outcomes.
- The partnership aims to generate meaningful real-world evidence (RWE) across all phases of the product lifecycle – introducing commercial expertise and insights far earlier in the clinical process – to
Read More
Aetion Closes $100M to Expand RWE Analytics Platform
What You Should Know:
- Healthcare technology company Aetion announced today a $110M Series C fundraise, led by Warburg Pincus, a leading global growth equity firm, with additional investments from B Capital and Foresite Capital. Aetion’s existing backers New Enterprise Associates (NEA) and Flare Capital Partners also joined the round. Aetion has raised a total of $212M to date.
- The company will use the new funds to extend the capabilities of its Aetion Evidence Platform, expand its
Read More
The Many Faces of AI in Clinical Trials
AI is being applied in several aspects for the clinical trial process today. From analyzing real-world data and scientific information to providing improved patient stratification and predictive outcomes, and assisting with different aspects of clinical trial operations. Here are some of the technologies using AI and machine learning in the clinical trial space today, outlining how they fit within the clinical trial ecosystem and discuss the impact on future clinical trial designs.
The
Read More
McKesson, Aetion Partner on Real-World Evidence (RWE) in Cancer Research
- Aetion and McKesson announced a strategic collaboration focused on advancing the use of real-world evidence (RWE) in cancer research. - Relationship pairs Aetion Evidence Platform and McKesson’s robust oncology data sets to accelerate regulatory-grade oncology studiesAetion and McKesson, today announced a strategic collaboration focused on advancing the use of real-world evidence (RWE) in cancer research to benefit patients, regulators, the biopharma industry, and payers. The partnership will
Read More
CB Insights Unveils ‘Digital Health 150’ List of Startups Transforming Healthcare
- CB Insights unveils first-ever annual ‘Digital Health 150’ list of digital health startups working to transform the healthcare industry.
- The Digital Health 150 companies span a wide spectrum of categories that involve all three key stakeholder groups for the healthcare industry — providers, payers, and patients.
- A total of 17 companies on the list are unicorns (private companies valued at $1B+). Of these, 12 companies are US-based, 3 are based in China, 1 is based in France, and 1
Read More
Health Tech VC Firm Flare Capital Partners Closes $255M Fund
Healthcare technology venture capital firm, Flare Capital Partners, today announced the closing of its second fund, Flare Capital Partners II, L.P., with a total of $255 million in committed capital. The oversubscribed fundraise follows up its inaugural fund, which was raised in April 2015, and establishes Fund II as one of the largest, independent, dedicated healthcare technology venture capital investment funds. Evolving Healthcare Industry Demands Innovative Business ModelsThe U.S. healthcare
Read More